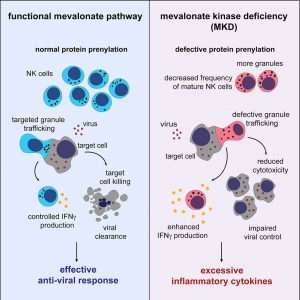
Rare genetic diseases often reveal biology we’d otherwise miss. One such condition, Mevalonate kinase deficiency, sits at the intersection of metabolism and immunity. A new study sheds light on how a defect in basic cellular chemistry can ripple outward to drive systemic inflammation (Figure 1).
MKD belongs to a group of disorders known as prenylopathies, where cells struggle to produce key lipid intermediates required for protein prenylation, process that anchors proteins to membranes and ensures proper intracellular signalling. While this biochemical defect has been known for some time, exactly how it leads to widespread immune dysregulation has remained unclear.
This work points to an unexpected culprit: dysfunctional Natural killer cells.
In both patients and mouse models, MKD was associated with a marked reduction in mature NK cells, the immune system’s frontline cytotoxic defenders against infected or abnormal cells. But the issue isn’t just numbers, it’s function.
Because prenylation is impaired, NK cells struggle with basic cellular logistics. One of the most critical defects lies in their inability to properly traffic cytolytic granules, the intracellular packages that contain perforin and granzymes used to kill target cells. Without efficient delivery of these granules, NK cells lose much of their killing function.
The result is a population of NK cells that are not only reduced but functionally compromised.
At first glance, fewer functional cytotoxic cells might suggest a weakened immune response. But MKD presents a paradox: alongside impaired cytotoxicity, there is heightened inflammation, particularly driven by elevated levels of Interferon gamma.
This imbalance likely stems from dysregulation rather than simple deficiency. With cytotoxic clearance impaired, immune activation becomes prolonged and poorly controlled. Instead of resolving infections efficiently, the immune system remains in a heightened state of activation, producing excess inflammatory cytokines.
This dysfunction becomes especially evident during viral challenge. In mouse models, MKD animals failed to effectively clear infections such as murine cytomegalovirus (MCMV). The impaired antiviral response was accompanied by:
- Persistent viral presence
- Elevated systemic IFN-γ
- Increased inflammatory tissue damage
In other words, the immune system is both ineffective and overactive at the same time—a hallmark of many autoinflammatory conditions.
One of the most clinically relevant findings is the potential to intervene downstream of this dysfunction. The study reports that treatment with a Janus kinase inhibitor helped reduce disease severity in a patient with severe MKD.
JAK inhibitors work by dampening cytokine signalling pathways, including those driven by IFN-γ. By interrupting this inflammatory cascade, they may help restore balance—even if the underlying metabolic defect remains.
Beyond MKD itself, these findings have wider implications. They highlight how tightly immune function is coupled to cellular metabolism and protein processing. When fundamental processes like prenylation are disrupted, the consequences extend far beyond biochemistry, reshaping immune cell development, function, and signalling.
Importantly, this work reframes MKD not just as a metabolic disorder, but as a disease of cytotoxic cell dysfunction and inflammatory imbalance.
As interest grows in targeting immune pathways for rare diseases, MKD offers a compelling example of how understanding cellular mechanisms can translate into therapeutic strategies. While JAK inhibition is not a cure, it represents a rational, mechanism-based approach to controlling disease.
Journal article: Munoz, M.A., et al. 2026. NK cell dysfunction and interferon gamma production underlie autoinflammation in mevalonate kinase deficiency. Immunity.
Summary by Stefan Botha