On April 25th 2022, we were treated with an interesting webinar by Professor Shane Crotty titled, “Adaptive Immunity and Immune Memory to SARS-CoV-2 after COVID-19.” Dr. Crotty’s lab studies the fundamental immunology underlying vaccine functions. Initially trained in molecular virology and then viral immunology, Crotty’s laboratory focuses on both the basic immunology of T follicular helper (Tfh) CD4 T cells (Science 2009, Immunity 2019), viral immunology including SARS-CoV-2 (Cell 2020), B cell immunodominance (Science 2016, Immunity 2018, Science 2019,), and the central roles of germinal centers and memory in vaccine immunology (Cell 2019, Nature Medicine 2020). Dr. Crotty was a Pew Scholar and a 2016-2019 Highly Cited Researcher. His recent COVID-19 immunology work has become the highest attention getting Cell paper ever (Cell 2020; Nature Reviews Immunology 2020).
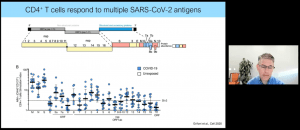
Dr. Crotty opened his talk with a brief introduction into the T-cell and antibody response during average COVID-19 infection (Figure 1). He highlighted the most important types of immunity during SARS-CoV-2 infection i.e. CD8 T killer cells, CD4 T helper cells and Antibodies from B cells. The aim of his team was to describe the immune response during COVID-19 infection. They found that CD4+ T cells respond to multiple SARS-CoV-2 antigens with the S (spike) protein being the most dominant target for both CD4+ and CD8+ T cells (Figure 2). This is especially important for the development of vaccines.
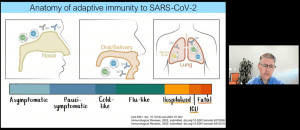
He then effectively established a rationale for investigating the mechanisms of protective immunity against COVID-19 (Figure 3). He highlighted how antibodies are correlate to CD4+ T cells, how the virus is susceptible to these neutralizing antibodies and how these antibodies can truly provide sterilizing immunity. Additionally, he described evidence of how T cell responses correlate with better outcomes following SARS-CoV-2 infection, followed by lower viral load.
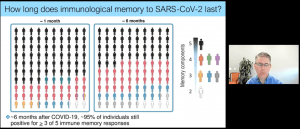
A team from his lab investigated the role of T cells in controlling SARS-CoV-2 infection, showing that T cells coordinate adaptive immunity which in turn is protective immunity and that age is a risk factor for shortcomings in adaptive immunity. He then described a follow-on study investigating how long immune memory to COVID-19 would last. They found that following 6 months from COVID-19 infection, 95% of individuals would still possess three or more out of five immune memory responses (Figure 4).
Further work described the effects of low-dose Moderna COVID-19 vaccine (RNA vaccine) on immune memory and described several factors that contribute to immune memory following vaccination. They also looked at reactive T cells in SARS-CoV-2 exposed and unexposed individuals, this was done through looking at the previously mentioned vaccine study (Figure 5). They measured this over time (0-6 months).
Finally, they looked at immune memory to the four COVID-19 vaccines and measured various immune cell types over time. They looked at antibodies, CD4+ T cells and CD8+ T cells. They showed that antibodies rose significantly in two doses of Pfizer and Moderna but declined after 6 months, whereby as for J&J the antibody level was low in the beginning but rose. The Novavax vaccine had high and stable antibody titer. The same could be said for the CD4+ T cell memory which looked very stable for all without the drastic decline as seen for the antibody titer for Pfizer and Moderna (only mild decline). For CD8+ T cells, the levels were high and increased over time for the vaccines, being lower for the J&J vaccine and Novavax vaccine when compared to Pfizer and Moderna.
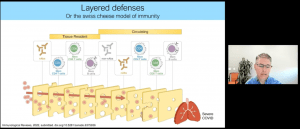
He closed with interesting work done in collaboration with other research groups, looking at tissue-localised immune memory as well as also providing us with a “Swiss cheese,” model describing how there are multiple layers of immune defense against COVID-19 infection especially when the individual is vaccinated (Figure 6).
Summary by Stefan Botha