A recent mechanistic study reveals how tumours disable key immune cells, and how restoring their metabolic fitness could dramatically improve cancer immunotherapy (Figure 1).
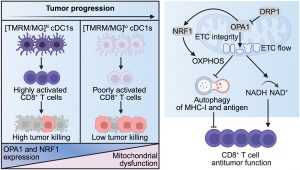
Figure 1: Mitochondrial metabolism and signaling orchestrate cDC1 antitumor responses.
(Left) [TMRM/MG]hi cDC1s more effectively prime CD8+ T cell responses and control tumor growth compared with [TMRM/MG]lo cells. Intratumoral cDC1s undergo mitochondrial dysfunction and decreased OPA1-NRF1 signaling during tumor progression. (Right) The OPA1-NRF1 axis maintains OXPHOS to inhibit autophagy and lysosome–mediated degradation of MHC-I and antigen. OPA1-mediated ETC flow also supports cDC1 immunogenicity by balancing the NAD+/NADH ratio. These effects are antagonized by DRP1. [Figure created with BioRender.com].
Researchers discovered that cDC1s within tumours exist in distinct mitochondrial states:
- High-functioning (polarizsed mitochondria) → strong ability to activate CD8+ T cells
- Low-functioning (depolarised mitochondria) → impaired immune activation
This metabolic distinction is critical. Only cDC1s with healthy, energy-producing mitochondria were able to effectively drive anti-tumour immune responses.
At the core of this process is the mitochondrial fusion protein OPA1, which regulates mitochondrial structure, energy production, and redox balance.
OPA1 works in concert with NRF1 to maintain oxidative phosphorylation (OXPHOS), the process by which mitochondria generate energy. This OPA1–NRF1 axis ensures that dendritic cells remain metabolically fit and immunologically active.
When OPA1 was deleted:
- Tumor growth increased
- CD8+ T cell responses were weakened
- Antigen presentation via MHC-I was impaired
The study uncovered several key mechanisms linking metabolism to immune activity:
- Sustained OXPHOS prevents degradation of MHC-I and tumour antigens, preserving the cell’s ability to present targets to T cells
- Proper mitochondrial function supports redox balance through NADH → NAD⁺ conversion
- Loss of mitochondrial fitness triggers autophagy and lysosomal degradation pathways that blunt immune activation
In contrast, the mitochondrial fission protein DRP1 counteracts these effects, promoting dysfunction.
As tumours progress, they actively push dendritic cells into a dysfunctional metabolic state:
- Reduced mitochondrial activity
- Lower OPA1–NRF1 signalling
- Fewer high-functioning cDC1s
This effectively shuts down one of the immune system’s most important anti-cancer pathways.
The therapeutic implications are substantial. When researchers introduced tumour antigen–loaded cDC1s with high mitochondrial activity into mice:
- Tumour control improved significantly
- Responses to Immune checkpoint blockade were enhanced
- Many mice completely rejected tumours
- Long-term immune memory was established
This confirms that metabolic fitness of dendritic cells is a limiting factor in immunotherapy success.
Together, these findings establish mitochondrial metabolism as a central regulator of dendritic cell function in cancer. Rather than focusing only on T cells, this work highlights the importance of optimizing upstream immune “orchestrators” like cDC1s.
Targeting mitochondrial pathways, particularly the OPA1–NRF1 axis, could provide a powerful strategy to:
- Overcome resistance to immunotherapy
- Enhance tumour antigen presentation
- Generate durable, long-lasting anti-tumour immunity
This research opens the door to metabolically engineered immune therapies, where boosting the energy capacity of immune cells becomes a key tool in the fight against cancer.
Journal article: You, J., et al. 2026. Mitochondrial metabolism and signaling direct dendritic cell function in antitumor immunity. Science.
Summary by Stefan Botha










