Endoperoxide artemisinin and its several derivates are significant role players in the treatment of malaria and are recommended as first-line drugs for artemisinin-based combination therapies by the World Health Organization. Delayed parasite clearance was found to be associated with reduced drug susceptibility of the ring stage as well as mutations in PFKELCH13, which encodes a kelch domain-containing protein on chromosome 13. PfKelch13 belongs to the top 5% of most conserved proteins in Plasmodium and comprises an N-terminal apicomplexan-specific region followed by a CCC domain, a BTB domain, and a six bladed kelch β-propeller domain. It has now become evident that mutations in PfKelch13 mutations remain a significant problem in our battle to eliminate malaria, an infectious disease that poses a significant problem to global health systems, especially in the developing world.
In a recent study, Schumann, et al., made use of selection-linked integration (SLI) in combination with glmS ribozyme-tagging and established a purification protocol for recombinant PfKelch13 to study the relevance of the abundance, conformational stability, and redox state of PfKelch13 for the artemisinin susceptibility in P. falciparum.
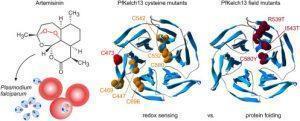
Figure 1: Schumann, et al., addressed the relevance of the redox and folding properties of the Keap1 homologue PfKelch13 for the artemisinin susceptibility of the malaria parasite Plasmodium falciparum (Schumann, et al., 2021).
In short, the researchers reported the down-regulation of PfKelch13 which results in ring-stage survival rates of up to 40% and that common field mutations have a destabilizing effect on the folding properties of PfKelch13. In addition, they reported that PfKelch13 exists in at least two different forms and established a protocol for the production of recombinant PfKelch13.
In their own words:
“In summary, in contrast to residues C469, C532, and C580, the surface-exposed thiol group of residue C473 appears to be essential. However, not the redox properties but impaired folding of PfKelch13, resulting in a decreased PfKelch13 abundance, alters the artemisinin susceptibility and is the central parameter for mutant selection.”
Journal article: Schumann, et al., 2021. Protein abundance and folding rather than the redox state of Kelch13 determine the artemisinin susceptibility of Plasmodium falciparum. Redox Biology.
Summary by Stefan Botha










