Macrophages are often described as key players in cancer, but that label hides an important truth: their role depends heavily on where they are and where they come from. A new study in lung cancer reveals a division of labour between macrophage subsets, showing how spatial organization within the tumour can determine whether these cells help fight cancer, or help it grow (Figure 1).
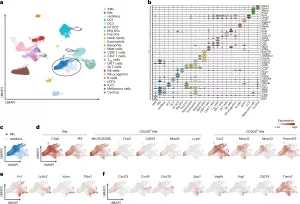
Figure 1: scRNA-seq of pulmonary melanoma identifies gene signatures in CD206hi IMs, CD206lo IMs and recMacs. a, Unbiased UMAP clustering of scRNA-seq data from flow-sorted extravascular CD64⁺CD11b⁺ mononuclear phagocytes isolated from the lungs of C57BL/6 WT mice on day 16 after intravenous injection of 4 × 105 B16F10 melanoma cells. Clusters include AMs, IMs, recruited macrophages (recMacs), dendritic cells 1 (DC1), dendritic cells 2 (DC2), inflammatory DCs (Inf DC2), migratory DCs (Mig DCs), pre-DCs, plasmacytoid DCs (pDCs), neutrophils, eosinophils, basophils, mast cells, CD8⁺ T cells, CD4⁺ T cells, regulatory T (Treg) cells, double-negative T (DN T) cells, γδ T cells, natural killer (NK) cells, NK progenitors, B cells, innate lymphoid cells (ILC2), melanoma cells and cycling cells. b, Key signature genes defining the unbiased clusters as in a. c, UMAP of Pf4+C5ar1+Mafb+ IMs and Ly6c2+Fn1+Vcan+ recMacs as in a. d, Feature plots showing the expression of C1qb and Pf4 in IMs (left), expression of Folr2, Cd163, Mmp9 and Lyve1 in CD206hi IMs (middle) and expression of Ccr2, Mmp12, Mmp13 and Tmem119 in CD206lo IMs (right). e, Feature plots showing expression for Fn1, Ly6c2, Vcan and Thbs1 in recMacs. f, Feature plots showing the expression of Cxcl13, Cxcl9 and Cxcl10 in CD206hi IMs (left) and Spp1, Vegfa, Arg1 and Cd274 in recMacs and Trem2 in recMacs and IMs (right).
Using single-cell and spatial transcriptomics, researchers mapped macrophages across the tumour landscape and identified two major populations. Tissue-resident interstitial macrophages (IMs), which originate locally, were found to behave very differently from recruited macrophages (recMacs), which arise from circulating monocytes and infiltrate the tumour.
At the edges of the tumour, particularly along bronchovascular regions, a subset of CD206^hi interstitial macrophages play a protective role. These cells produce chemokines such as CXCL9, CXCL10, and CXCL13, which are essential for recruiting lymphocytes and organizing them into structured immune niches. In doing so, they promote the formation of tertiary lymphoid structures, sites where immune responses can be initiated and sustained locally. This positioning allows them to act as coordinators of antitumor immunity, helping to bring immune cells into the fight and supporting tumour control.
Deeper within the tumour, however, the picture changes. Another subset of interstitial macrophages produces CCL2, a chemokine that attracts monocytes from the bloodstream. These monocytes differentiate into Ly6c2⁺Fn1⁺Vcan⁺ recruited macrophages, which contribute to an immunosuppressive environment. Instead of supporting immune attack, this pathway reinforces tumour survival by dampening effective immune responses. This dynamic is a core feature of the tumour microenvironment, where cellular interactions and spatial context shape disease progression.
The study also extends beyond the tumour itself, uncovering an unexpected role for immune cells in tumour-draining lymph nodes. Here, monocyte-derived dendritic cells (moDCs) act as suppressive antigen-presenting cells, limiting the effectiveness of immune activation. Importantly, this suppressive circuit can be disrupted. By blocking CCR5, a receptor involved in immune cell migration, with the drug maraviroc, researchers were able to prevent these moDCs from trafficking to lymph nodes. When combined with neoantigen vaccination, this intervention enhanced dendritic cell–mediated antitumor immunity, leading to stronger immune responses.
Together, these findings highlight a more nuanced view of macrophages in cancer. Rather than being broadly pro- or anti-tumour, macrophages exist along a spectrum defined by their lineage and location. Some subsets actively support immune surveillance and tumour control, while others establish pathways that tumours exploit to evade immunity.
Journal article: Ghosh, S., et al. 2026. Chemokine-defined macrophage niches establish spatial organization of tumor immunity. Nature Immunology.
Summary by Stefan Botha










