The US National Institutes of Health (NIH) hosts regular COVID-19 Scientific Interest Group meetings. This week we highlight the 16th COVID-19 SIG meeting that featured a talk by Prof. Paul Bieniasz*Ph.D (Rockefeller University) on “Neutralizing Antibodies to SARS-CoV-2“.
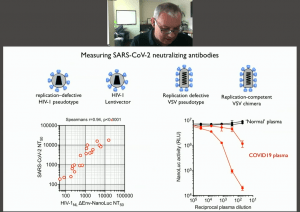
He began his talk describing the use of high-throughput neutralisation antibody (nAb) assays to detect the variability of the neutralisation capacity of nAbs in convalescent plasma. Further, he showed that the half-life of nAbs is low (approximately 1-month) and can decrease to a tenth of the level detected at baseline in 6 months. He then described the identification of potent monoclonal nAbs from B cell clones isolated from convalescent sera. He showed that potent nAbs had a low degree of somatic hypermutation. He presented findings that demonstrated that these potent nAbs have potential sterilising effect in vivo using a mouse-adapted SARS-CoV-2 pseudovirus model.
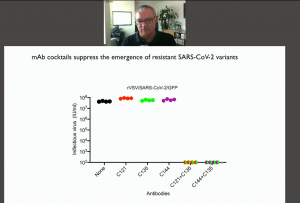
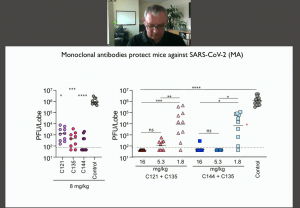 Selection of Ab resistant-SARS-CoV-2 viral strains presents a potential challenge of monoclonal Ab or convalescent therapy. In the final part of his talk, Prof Bieniasz described in vitro experiments that were used to assess the development of Ab resistance which posses potential challenge in the clinical setting. They developed a replication-competent VSV/SARS-Cov-2 chimera that recapitulates the neutralising properties of the original SARS-Cov-2 backbone and enabled the selection Ab resistant virus. Further, using this approach they were able to determine which combination of mAbs could be used to prevent the selection of Ab resistance virus.
Selection of Ab resistant-SARS-CoV-2 viral strains presents a potential challenge of monoclonal Ab or convalescent therapy. In the final part of his talk, Prof Bieniasz described in vitro experiments that were used to assess the development of Ab resistance which posses potential challenge in the clinical setting. They developed a replication-competent VSV/SARS-Cov-2 chimera that recapitulates the neutralising properties of the original SARS-Cov-2 backbone and enabled the selection Ab resistant virus. Further, using this approach they were able to determine which combination of mAbs could be used to prevent the selection of Ab resistance virus.
*Dr. Bieniasz research groups conducts research on how host genes influence the replication of viruses, with an emphasis on human and primate immunodeficiency viruses. They are also now conducting a variety of investigations into the nature of innate and adaptive immunity to SARS-CoV-2, including the development of techniques to identify protective antibodies and therapeutics. (Source: NIH Videocast Website)
Summary by Cheleka AM Mpande