Plasmacytoid dendritic cells (pDCs) are best known as the immune system’s rapid responders, cells that can unleash large amounts of type I interferons during viral infection. But that simple description hides a much more complex reality. A new study using single-cell transcriptomics shows that pDCs are far from uniform (Figure 1). Instead, they exist in a spectrum of functional states shaped by where they reside and how early in life they develop.
What emerges is a picture of pDCs as highly adaptable cells, tuned by both tissue environment and developmental timing.
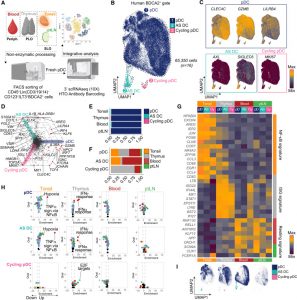
By profiling pDCs from blood, thymus, lymph nodes, and tonsils across fetal, infant, and paediatric stages, researchers uncovered distinct transcriptional programs linked to specific tissues.
In the thymus, pDCs were strongly imprinted by Type I interferon signalling, suggesting a role in antiviral readiness early in immune development. In contrast, tonsillar pDCs showed enrichment for NF-kappa B–driven programs, aligning with their role in mucosal immunity where constant immune activation is the norm. Meanwhile, pDCs circulating in blood and lymph nodes appeared comparatively “resting,” maintaining a more quiescent baseline state.
Rather than a single fixed identity, pDCs seem to adopt context-dependent roles, adjusting their function to local immune demands.
One of the more intriguing findings is the identification of a pDC subset present across tissues that produces prostaglandin D2 and responds to dopamine. This points to a potential interface between the immune and nervous systems—an emerging area of biology where immune cells are influenced by neurotransmitters.
The idea that pDCs might integrate neuromodulatory signals suggests they could play roles beyond classical immunity, possibly linking inflammation, tissue signalling, and even behaviour in ways that are only beginning to be understood.
Developmental timing also plays a major role in shaping pDC biology. In fetal and infant tissues, a population of actively dividing, or “cycling”, pDCs is abundant, particularly in lymphoid organs. These cells gradually decline with age but remain consistently enriched in the bone marrow throughout life.
This persistence hints at a long-lived reservoir of proliferative pDCs, suggesting that early-life immune development may leave behind cellular populations that continue to influence immunity well into adulthood.
This developmental insight becomes especially relevant in disease. In Blastic plasmacytoid dendritic cell neoplasm, a rare and aggressive malignancy, the study found an accumulation of these cycling pDCs within the bone marrow, particularly those carrying mutations.
This suggests that the bone marrow niche may act as a reservoir for malignant transformation, where long-lived, proliferative pDCs acquire genetic alterations and expand into cancer.
Taken together, these findings challenge the traditional view of pDCs as a single-function interferon-producing cell type. Instead, they appear to be:
- Developmentally programmed across early life
- Functionally specialized by tissue environment
- Potentially influenced by neuroimmune signals
- Implicated in disease through long-lived proliferative subsets
The study reframes pDCs as dynamic, context-aware immune cells, capable of adapting their roles across both space and time.
Understanding this diversity has practical implications. It may help explain why pDCs behave differently in infections, autoimmune diseases, or cancer and why targeting them therapeutically has produced mixed results.
Journal article: Antonova, A.U, et al. 2026. Age- and tissue-dependent diversity of human plasmacytoid dendritic cells uncovers a cycling subset dominant in early life and cancer. Immunity.
Summary by Stefan Botha