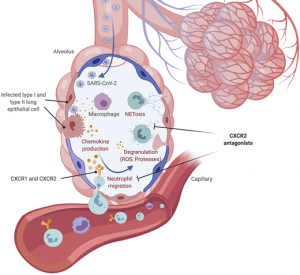
Infiltration and activation of neutrophils into SARS-CoV-2–infected lungs. Infected lung epithelial cells and inflammatory innate immune cells such as macrophages release the leukocyte attracting chemokines CXCL1, CXCL2, CXCL5, and CXCL8. Neutrophils are recruited and activated by these chemokines via their receptors CXCR1 and CXCR2. Subsequent release of antimicrobial effectors, such as ROS, proteases, and NETs, causes severe collateral damage in the lung. CXCR2 antagonists may inhibit lung damage by blockade of neutrophil migration and activation. Created with BioRender. (Source: Koenig et al., 2020 JEM)
With recent results published from the RECOVERY trial showing that dexamethasone reduces 28-d mortality among patients receiving invasive mechanical ventilation or oxygen revealed important understanding of the inflammatory mechanisms leading to severe COVID-19. Corticosteroid treatment could be regarded as a “heavy approach” to relieving the devastating effects of acute respiratory distress syndrome and would potentially inhibit the signalling pathways leading to robust T and B cell immunity. Perhaps then a more selective approach could be used to target the now known inflammatory overreaction leading to severe disease and mortality. A Viewpoint in the July 17 2020 edition of JEM highlights the possible use of CXCR2 antagonists to block the homing of neutrophils, and other inflammatory myeloid cells, into infected lungs and other tissues. The authors discuss the evidence of neutrophil involvement in COVID-19 and the effectiveness of CXCR2 inhibition in preclinical models.
Journal Article: Koenig et al., 2020. Blocking inflammation on the way: Rationale for CXCR2 antagonists for the treatment of COVID-19. JEM
Summary by Clive Gray










