The development of therapeutics for immune-related disorders often involves identifying molecules responsible for immune dysregulation and targeting them using drugs. This type of immune therapy is also used for cancer treatment, owing to the important role that immune cells play in cancer progression. One such type of a potential therapeutic target is an “immune checkpoint” protein called PD-L2, which is expressed by dendritic cells (DCs) and macrophages. PD-L2 plays various important functions in the regulation of different types of immune cells, including the suppression of T cells. Not surprisingly, this molecule has been extensively explored as a target protein for immunopathologies including cancer. But, despite years of research, the exact mechanism on how this protein gets regulated has remained unclear until now.
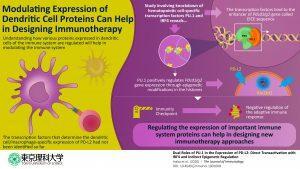
Recently, a group of researchers at Tokyo University of Science, led by Professor Chiharu Nishiyama“ investigated the gene expression mechanism of PD-L2, a cell-surface molecule that is specifically expressed by DCs and macrophages.’’ In DCs, PD-L2 acts as a “checkpoint” that ensures that the immune cells do not incorrectly destroy healthy cells. Because the gene “Pdcd1lg2” is responsible for coding the PD-L2 protein, the scientists wanted to understand how this specific gene is regulated. Prof Nishiyama explains, “Previous studies had shown that the artificial expression of PU.1 on non-dendritic immune cells transformed these cells into DCs. This made us wonder if PU.1 had any role in the expression of PD-L2, one of the prominent surface proteins of DCs.”
To begin with, the scientists studied the effect of “knocking down” PU.1 and a few other proteins. They found that in the absence of PU.1 and its associate molecule IRF4, the expression of PD-L2 was severely compromised. Once it was clear that PU.1 and IRF4 were involved in regulating the expression of PD-L2, the scientists carried out subsequent experiments using techniques such as “chromatin immunoprecipitation” to know exactly how this happens. They found out that PU.1 and IRF4 together bind to a region on the DNA called the “EICE sequence” and stimulate the expression of Pdcd1lg2 gene. It also became clear that PU.1 indirectly plays a role in epigenetic modification, specifically histone acetylation*
In sum, this study sheds light on the molecular events that take place in the regulation of the PD-L2 protein. This paves the way for further development of drugs that can regulate this protein in immune disorders. Prof Nishiyama concludes, “We believe that our findings have remarkable potential to improve the therapeutic approach against immune system-mediated diseases.”
Reference: Inaba et al., 2020. Dual Roles of PU.1 in the Expression of PD-L2: Direct Transactivation with IRF4 and Indirect Epigenetic Regulation. The Journal of Immunology
Summary provided by Indrani Das
About The Tokyo University of Science
Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan’s development in science through inculcating the love for science in researchers, technicians, and educators. Website:
About Professor Chiharu Nishiyama from Tokyo University of Science: Professor Chiharu Nishiyama is affiliated with Faculty of Industrial Science and Technology, Department of Biological Science and Technology. She received her Bachelor’s, Master’s, and PhD degree from The University of Tokyo in 1988, 1990, and 1997, respectively. Her main areas of research include applied biochemistry, immunology, allergy, molecular biology, transcription factor, and cell development. Prof Nishiyama was awarded the JSBBA Award for Young Scientists in 2005. Till date, she has around 115 research papers to her credit.
Media contact regarding original article: Tsutomu Shimizu (mediaoffice@admin.tus.ac.jp)