In a recent study, scientists discovered pathways that may be therapeutically targeted to enhance patient outcomes and have described a kind of immune cell that causes chronic organ transplant failure in a mouse model of kidney transplantation (Figure 1).
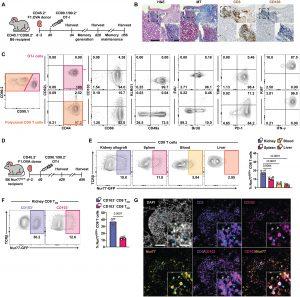
Figure 1: TRM cells form in renal allografts and encounter local cognate antigen. (A) (Balb/c × B6.OVA) F1 (F1.OVA) kidney allografts were transplanted into B6 recipients followed by adoptive transfer of 1 × 106 effector OT-I cells 2 days after transplantation. Transplant recipients were harvested at indicated times. (B) H&E-, MT-, anti-CD3– and anti-CD103–stained sections of renal allograft tissue 56 days after transplantation. Insets highlight areas of T cell infiltration. Blue in MT staining indicates fibrosis. Whole image scale bar, 200 μm. Inset scale bar, 50 μm. n = 6 mice. (C) Transferred CD90.1/90.2+ OT-I and recipient-derived polyclonal CD90.2+ CD8 T cells from 28-day after transplant kidney allografts were analyzed for TRM markers after gating on extravascular graft CD8+ T cells as shown in fig. S1B. BrdU water was administered 7 days before and EdU was injected 1 hour before harvest. n = 8 mice. (D) F1.OVA kidney allografts were transplanted into Nur77-GFP B6 recipients followed by adoptive transfer of 1 × 106 effector OT-I cells 2 days after transplantation. Transplant recipients were harvested on days 28 and 56. n = 4 mice per time point. (E) Representative plots (left) and percentages (graph, right) of Nur77-GFP expression by CD8 T cells in various tissues on day 56. (F) Representative plots (left) and percentages (graph, right) of Nur77-GFP expression based on CD103 expression. (G) Immunofluorescence staining of renal allograft tissue on day 56 for DAPI (white), CD3 (purple), CD103 (magenta), and Nur77 (yellow). Blue arrows point to cells coexpressing CD3, CD103, and Nur77. Green arrows point to cells expressing only CD3. Whole image scale bar, 100 μm. Inset scale bar, 20 μm. n = 5 mice. P values were determined by (E and F) two-tailed unpaired t test.
They have previously demonstrated that tissue-resident memory T cells, a subset of immune cells, was responsible for persistent rejection. Tissue-resident memory T cells exist inside organs, as opposed to the majority of memory T cells, which circulate in the blood.
Researchers found two elements in this study that sustain tissue resident memory T cells in kidney transplants over time. The initial factor is the antigen itself, which are the chemicals used by T cells to identify the donor graft as being foreign. Residents T cells are always exposed to such antigens since they live within the kidney transplant. A cytokine, or inflammatory signalling protein, by the name of IL-15, is the second component.
Dendritic cells, a different class of immune cell that captures both the antigen and IL-15 and presents them to receptors on resident memory T cells, are also essential to this process. Researchers observed a decrease in resident memory T cell number and functioning when dendritic cells were reduced or prevented from presenting antigen or IL-15.
The findings, in the researchers’ opinion, may pave the way for clinical studies to test the antibody in transplant recipients in an effort to reduce chronic rejection.
Journal article: Tieu, R., et al., 2023. Tissue-resident memory T cell maintenance during antigen persistence requires both cognate antigen and interleukin-15. Science Immunology.
Summary by Stefan Botha










