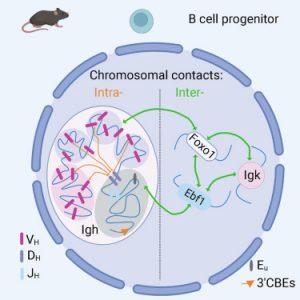
In the field immunology, understanding the arrangement of antibody variation is one we have yet to understand. A recent study has shed light on the intricate DNA associations within B cells, revealing the heart of antibody diversity (Figure 1).
Researchers uncovered a fascinating facet of B cell biology. They found that the 3D configuration of DNA in these cells allows genes situated far apart to converge during antibody generation, yielding the diverse arsenal needed for robust disease protection. Astonishingly, each B cell exhibits a unique folding pattern for this part of the genome, eschewing a common template. This implies an array of infinite possibilities for combining genes in distinctive sequences.
The research aimed to clarify the mechanisms underpinning antibody diversity. While the cell uses cutting and pasting from a variety of antibody gene mechanisms, an interesting finding remained – distant genes were utilized as frequently as nearby ones. This implied an intricate coordination mechanism ensuring all necessary elements were readily available.
The research team pioneered a next-generation sequencing method, an evolution of the enriched Hi-C genome analysis, to unveil deeper DNA configuration insights within B cells. This technique enabled the creation of 3D simulations for thousands of gene structures, validating their discoveries.
Additionally, the study unveiled that antibody genes closely neighboured a small subset of genes on different chromosomes. Upon characterization, these genes were linked to B cell development.
This research reveals the complex genetic dance underlying antibody diversity. It not only expands our comprehension of immunology but also sets the stage for potential breakthroughs in disease protection and treatment through tailored antibody therapies.
Journal article: Mielczarek, O., et al., 2023. Intra- and interchromosomal contact mapping reveals the Igh locus has extensive conformational heterogeneity and interacts with B-lineage genes. Cell Reports.
Summary by Stefan Botha