For decades, scientists have observed a strong link between Epstein–Barr virus (EBV) infection and autoimmune diseases such as systemic lupus erythematosus (SLE). Yet the underlying mechanism, how a common virus could contribute to a complex, systemic autoimmune disorder, has remained elusive. A new study now provides evidence that EBV may not just be associated with lupus but could actively help drive it (Figure 1).
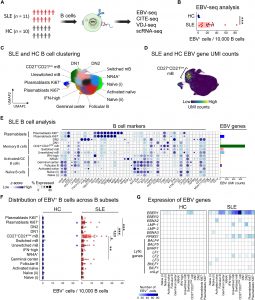
Figure 1: EBV-seq enables identification of EBV-infected B cells. (A) An EBV-seq primer design was incorporated into a 10x Genomics bead-based scRNA-seq workflow that included CITE-seq performed using oligonucleotide-barcoded Abs specific for the prototypical markers for B cell subsets. BCR sequencing was performed by analysis of the expressed variable diversity and joining (VDJ) immunoglobulin genes, followed by analysis of immunoglobulin heavy (H) and light (L) chain gene expression. (B) Shown is the EBV genome annotated with the EBV genes (blue font) and small noncoding RNAs (green font) for which primers were included in EBV-seq. (C) Comparison of standard 10x Genomics scRNA-seq with EBV-seq 10x Genomics scRNA-seq for detection of EBV+ B cells in a PBMC sample from a patient with SLE; EBV UMI counts are represented by intensity of brown dots. ADT, antibody-derived tag.
At the centre of SLE is a breakdown in immune tolerance, leading to the production of antibodies against the body’s own nuclear components. These so-called antinuclear antibodies are a hallmark of the disease. What this study reveals is that EBV may directly interfere with this balance by targeting and reprogramming a very specific subset of immune cells: autoreactive B cells.
Using a single-cell RNA sequencing approach designed to detect EBV-infected cells, researchers found that infected B cells are fundamentally different from their uninfected counterparts. Rather than behaving like typical antibody-producing cells, these EBV-positive B cells adopt features more commonly associated with antigen-presenting cells. In other words, they begin to actively instruct other parts of the immune system, particularly T cells.
This shift appears to be driven by a viral protein, EBNA2, which binds to key regulatory regions in the host genome and rewires gene expression. The affected genes include those involved in B cell activation and antigen presentation, effectively pushing these cells into a more inflammatory, immune-stimulating state. This process reflects a broader principle of viral manipulation of host immunity, where pathogens hijack cellular machinery to their advantage.
Crucially, these reprogrammed B cells are not random, they are enriched for cells that already recognize self-antigens. The study confirmed that antibodies produced by these EBV-infected B cells bind to classic lupus-associated nuclear targets, reinforcing their role in autoimmunity. Once activated, these cells can present self-antigens to helper T cells, initiating a cascade of immune activation. These T cells, in turn, stimulate additional autoreactive B cells.
This creates a self-propagating loop within the systemic lupus erythematosus immune network. What begins as a localised viral effect in a subset of B cells can escalate into widespread immune dysregulation, ultimately driving systemic disease.
The findings offer a mechanistic explanation for the long-suspected link between EBV and lupus. Rather than acting as a general immune trigger, EBV appears to selectively infect and reprogram autoreactive B cells, converting them into potent drivers of immune activation. This targeted effect helps explain why EBV is associated with specific autoimmune diseases rather than causing broad immune dysfunction.
Beyond lupus, the implications are significant. EBV has also been implicated in conditions such as multiple sclerosis and Long COVID, raising the possibility that similar mechanisms of immune reprogramming may be at play across different diseases.
By uncovering how a common virus can reshape immune cell identity and function, this study provides a new framework for understanding autoimmune disease development. autoreactive immune circuits.
Journal article: Younis, S., et al. 2025. Epstein-Barr virus reprograms autoreactive B cells as antigen-presenting cells in systemic lupus erythematosus. Science Translational Medicine.
Summary by Stefan Botha










