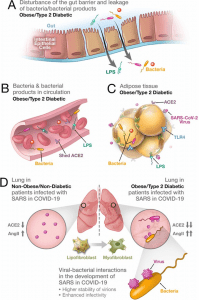
Synergistic interaction between SARS-CoV-2 and bacteria/bacterial products as a possible reason for more severe forms of COVID-19 in patients with obesity and T2D. (A) Metabolic dysregulation in obese/T2D patients provides the conditions for disturbance of the gut barrier and leakage of bacteria/bacterial products into the circulation. This dysregulation can be additionally enhanced through viral-induced disbalance in a local renin-angiotensin system (RAS). (B) Leakage of bacteria and bacterial products into the circulation provides their system-wide dissemination. (C) In the setting of a synergistic effect of viral–bacterial interactions, some bacterial products can trigger an intense response in the adipose tissue. In obesity and T2D, bacteria and bacterial DNA have been found as long-term constituents in different fat depots. (D) Increase in circulating LPS will lead to the accumulation of endotoxins in the lung, causing progressive pulmonary inflammation and vascular complications. Viral–bacterial interactions lead to hypercytokinemia – the dysproportional increase in the expression of pro-inflammatory cytokines, which is much higher than what an individual exposure to either viral or bacterial agents can achieve. This also leads to higher stability of virions and enhanced infectivity of SARS-CoV-2. A local synergistic pulmonary ACE2 deficiency develops a disbalance between vasodilating and vasoconstricting RAS agents resulting in inflammation. This interaction can also cause enhanced trans-differentiation of lipofibroblasts into myofibroblasts in obese and T2D patients causing the pulmonary fibrosis. (Source: )
A recent review in eLife synthesises data from multiple studies to propose a synergistic interaction between SARS-CoV-2 infection and obesity and/or diabetes leading to impaired endothelial function and gut barrier function. This impairment results in microbial translocation into the systemic circulation and the ensuing cytokine storm. Adipocytes richly express ACE2 and are hence an easy target for SARS-CoV-2 infection and the establishment of a viral reservoir. ACE2 may also be shed from adipose tissue into the systemic circulation and deposited in the lungs and thus “modifying pulmonary susceptibility to SAR-CoV-2 infection”. Paradoxically, such an acute viral infection causes a reduction in ACE2 expression in other organs and tissues leading to ACE2 deficient expression elsewhere. This causes a disturbance of the ACE2:renin-angiotensin system (RAS). This can result in increased plasma levels of angiotensin II (Ang II), triggering vasoconstrictive responses. Ang II levels have shown correlations with the severity of lung injury in patients with severe COVID-19 and these disturbances are exacerbated in patients with obesity and diabetes. This injury can result in lung dysbiosis and bacteria and/or their products in the lungs promotes an additional lung injury resulting in “synergistic interaction between viral and bacterial pathogens”.
Summary by Clive Gray










