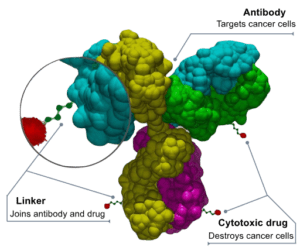
Antibody-drug conjugates (ADCs) are therapeutics which combine drugs with highly antigen-specific antibodies. Currently, most of these conjugates are formed through the introduction of mutations or chemical approaches. These usually lead to heterogeneity, instability and efficacy problems. Researchers, led by Christoph Rader, have now developed a conjugation method which is independent of mutations but rather manipulates a natural lysine residue on the antibody. Their novel ADCs were effective at fighting three different types of cancers both in vitro and in vivo.
Currently, four types of ADCs have been FDA-approved and approximately 60 are currently in preclinical trials. However, the way many ADCs are made results in heterogeneous mixtures which would lower the efficacy of the treatment. This has led researchers to develop novel strategies which reduce this heterogeneity such as incorporating certain mutations and using certain chemical conjugation methods. However, these strategies are multiple step processes which may also need long reaction times.
Therefore, the researchers developed a more efficient method of designing ADCs based on a one-step conjugation of the drug to a natural lysine residue on an antibody called h38C2. This antibody is a humanized anti-hapten antibody which contains the lysine of interest in a hydrophobic area, making the lysine highly nucleophilic at physiological pH. The h38C2 antibody recognizes cancer-specific antigens and therefore would be a good candidate for a cancer immunotherapy.
The researchers found that their conjugation method was very robust, resulting in homogenous mixtures of the ADCs. They tested the ADCs in cancer cells lines and in vivo cancer mouse models. The researchers found that their ADCs were effective against breast cancer, multiple myeloma and non-Hodgkin lymphoma.
In summary, researchers have developed a novel strategy for the development of ADCs against cancer. This strategy is easier and faster than previous methods and may aid in the rapid development of therapies towards different types of cancers.
Journal article: Nanna et al., 2017. Harnessing a catalytic lysine residue for the one-step preparation of homogeneous antibody-drug conjugates. Nature Communications
Article by Thandeka Moyo