A new study has found encouraging results from a small trial of an individualized neoantigen mRNA vaccine in patients with triple-negative breast cancer (TNBC), a subtype known for high relapse rates even after early-stage treatment (Figure 1).
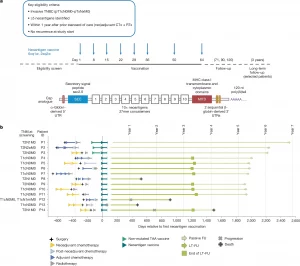
Figure 1: Trial design, RNA vaccine design and clinical course of patients with TNBC. a, Top, clinical trial design. Patients with early TNBC after surgery and neoadjuvant and adjuvant chemotherapy (CTx), with or without radiotherapy (RTx), were eligible within one year after having received standard of care treatment. Middle, a run-in cohort (n = 3, intra-patient dose escalation: 14.4 µg, 29 µg and 50 µg, followed by 5 × 50 µg) preceded the main cohort (n = 12) with patients dosed with 8 × 50 µg mRNA–LPX vaccine. Patients in the run-in cohort received pre-treatment (off-the-shelf mRNA–LPX vaccine assembled according to the tumour expression profile of the patient from a pre-manufactured warehouse of eight non-mutated TAAs) as a bridge until their personalized vaccine was available. Bottom, the individualized neoantigen mRNA vaccine consisted of 20 neoantigen vaccine targets encoded on two poly-lipoplexed mRNA molecules. For SNVs, the 27mer peptide region with the changed amino acid in the centre was used, and for frameshift-inducing insertions or deletions the sequence from the changed amino acid to the next stop codon was used. Open reading frames encoding the neoantigen target concatemers were flanked by a secretory signal peptide (SEC) and an MITD for enhanced HLA presentation. The 5′ and 3′ untranslated regions (UTRs) and the poly(A) tail designs of mRNAs were optimized for stability and translational efficiency. The three patients pretreated with bridging TAA vaccine were followed up passively. b, Prior treatment, trial regimen and disease course of patients enrolled in this trial. Out of 15 consented patients, 1 was discontinued after 3 vaccinations owing to TEAEs (hypotension, grade 3; nausea, grade 2; chills, grade 1) and is not displayed here. FU, follow-up; LT-FU, long-term follow-up; q1w, once a week; q2w, twice a week; TNM, tumour, nodes and metastasis grading system.
Fourteen patients received a personalized vaccine designed to target tumour-specific neoantigens following surgery and standard therapy. In nearly all participants, the vaccine generated strong T cell responses against multiple tumour mutations. These responses were largely newly induced rather than boosted from prior immunity and remained detectable and functional for several years.
Detailed immune profiling showed that vaccine-activated T cells differentiated into two key populations: cytotoxic effector cells poised to attack tumour cells, and stem-like memory T cells capable of sustaining long-term immunity. Notably, 11 of the 14 patients remained relapse-free for up to six years after vaccination.
Three patients experienced recurrence, offering insight into potential immune escape mechanisms. One patient with the weakest vaccine response relapsed but later achieved complete remission with anti-PD-1 therapy. Another developed tumour lacking MHC class I expression, allowing cancer cells to evade T cell recognition. The third patient, who carried a BRCA mutation, relapsed with a genetically distinct tumour.
Overall, the study demonstrates that personalized mRNA vaccines are feasible in TNBC and can induce durable, functional anti-tumour T cell responses. The findings also highlight potential pathways of resistance that could inform future vaccine and combination immunotherapy strategies.
Journal article: Sahin., U. et al. 2026. Individualized mRNA vaccines evoke durable T cell immunity in adjuvant TNBC. Nature.
Summary by Stefan Botha










