Our blood is constantly renewed by rare but powerful hematopoietic stem cells (HSCs) tucked deep inside the bone marrow. These stem cells are the wellspring of our immune and blood systems, giving rise to everything from red blood cells to infection-fighting white blood cells. But as we age, or when diseases like leukemia wreak havoc, the regenerative powers of these HSCs begin to falter. What drives this change at the cellular level?
A new study has mapped out the molecular and metabolic transformations that blood stem cells undergo during aging, differentiation, and disease (Figure 1). For the first time, scientists have combined transcriptomics, metabolomics, and lipidomics in an integrated analysis of human HSCs, despite their extreme rarity.
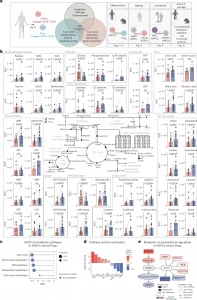
Figure 1: Metabolic profile of human BM HSPCs and their downstream progenitors. a, The experimental study design depicting the generated datasets and their respective figure numbers. Created with BioRender.com. b, A schematic summary of the metabolomics results per pathway in human HSPCs and progenitors. Quantification bar plots as mean ± standard deviation show raw intensity values per biological replicate. Paired samples are linked by grey lines. Differentially abundant metabolites are depicted as coloured dots in the scheme. n = 17. P-adjusted value after paired two-sided Student’s t-test or Wilcoxon test is shown. NS, not significant; ND, not detected. c, GSEA of metabolic pathways on RNA-seq of human HSPCs versus progenitors. n = 4. d, Pathway activity score estimated by decoupleR in HSPCs versus progenitors RNA-seq. Differentially activated pathways are shown (P < 0.05). e, A subnetwork representing the COSMOS mechanistic hypothesis of metabolic and transcriptomic regulation of choline pathway, based on HSPCs versus progenitors metabolomics and RNA-seq datasets. Each node represents a gene or metabolite. The border colour depicts the estimated activity by the model. The arrow shape corresponds to the type of regulation based on ground knowledge. The fill colour of elements shows their level of differential expression or abundance in our measured data. P-adjusted value; *P < 0.05, **P < 0.01, ***P < 0.001. In b and c, n indicates the number of biological replicates per condition. For b, ten independent experiments were performed. NAA, N-acetylaspartic acid; α-KG, alpha-ketoglutarate; PEP, 2-phosphoenolpyruvate; GSSG, glutathione disulfide.
Using advanced FACS sorting and low-input metabolic profiling, the team discovered that:
- Healthy HSCs are metabolically quiet, conserving energy and resources to protect their long-term function.
- As HSCs differentiate into progenitor cells, their metabolism ramps up, preparing for active cell division and specialization.
- In both aging and leukemia, this metabolic profile shifts dramatically, reflecting changes that may impair stem cell function.
One key player stood out: choline, a nutrient found in foods like eggs and soy. High levels of choline were seen in healthy, youthful HSCs, but levels dropped sharply with aging and disease.
The study also explored how changes in lipid composition affect the cell membrane architecture, altering how HSCs sense and respond to their environment. This aspect is crucial, as membrane dynamics can influence how stem cells receive signals to divide, specialize, or stay dormant.
The discovery that nutrient signals like choline can preserve stem cell identity offers exciting new possibilities.
By mapping the molecular signatures that define youth, aging, and disease in human blood stem cells, this study offers more than insight, it opens a new chapter in stem cell biology, with real therapeutic promise for combating blood disorders, enhancing recovery, and supporting healthy aging.
Journal article: Maria-Eleni Lalioti., ME., et al, 2025. Differentiation, ageing and leukaemia alter the metabolic profile of human bone marrow haematopoietic stem and progenitor cells, Nature Cell Biology.
Summary by Stefan Botha










