Malaria Life-Cycle
- Malaria, an intracellular parasite, is of widespread importance in the world
- When an infected Anopheles mosquito bites a human, it injects sporozoites into the small blood vessels.
- Sporozoites migrate to the liver where they infect hepatocytes, after which the parasite develops into a multinucleate liver stage (schizont) that contains merozoites (Figure 1).
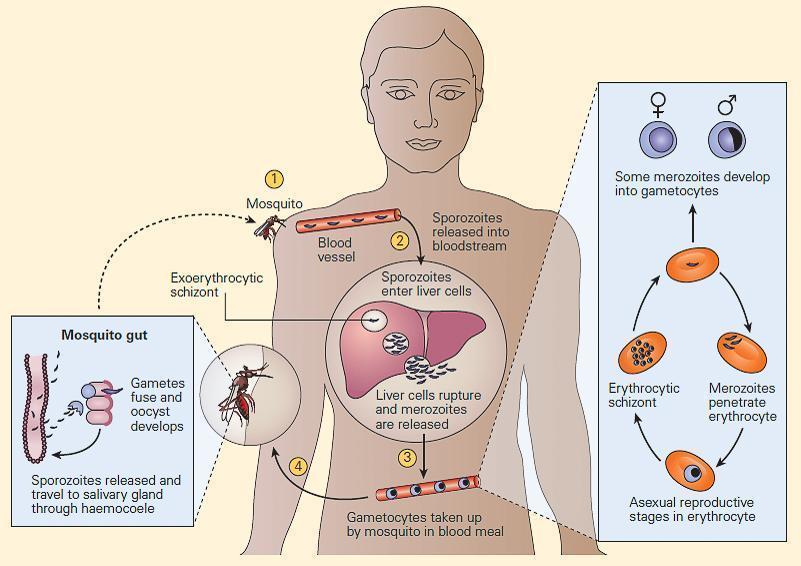
Figure 1. The life cycle of Plasmodium falciparum in the human host and mosquito vector. The mosquito injects sporozoites into the host (1), which are carried through the blood to the liver (2), where they invade hepatocytes and undergo a process of asexual (mitotic) replication to give rise to an exoerythrocytic schizont. Up to this point, the infection is nonpathogenic and clinically silent. After about seven days, the liver schizonts rupture to release many thousands of merozoites into the blood (3). Each merozoite invades an erythrocyte and divides mitotically to form an erythrocytic schizont containing up to 20 daughter merozoites (right, inset). These merozoites can reinfect fresh erythrocytes, giving rise to a cyclical blood-stage infection with a periodicity of 48 to 72 hours, depending on the Plasmodium species. As-yetunknown factors trigger a subset of developing merozoites to differentiate into male and female gametocytes, which, when taken up by a feeding mosquito, give rise to extracellular gametes (4). In the mosquito mid-gut, the gametes fuse to form a motile zygote (ookinete), which penetrates the mid-gut wall and forms an oocyst, within which meiosis takes place and haploid sporozoites develop (left, inset). (Reproduced with permission from Stevenson MM, Riley EM. Innate immunity to malaria. Nat Rev Immunol. 2004;4:169-80.) [Reproduced with permission from Bellanti, JA (Ed). Immunology IV: Clinical Applications in Health and Disease. I Care Press, Bethesda, MD, 2012].
- This stage is referred to as exoerythrocytic schizogony and may occur with a single infecting parasite (e.g., P. falciparum) or with multiple organisms (e.g., P. vivax, Povale).
- The mature schizonts eventually rupture, releasing thousands of uninucleate merozoites into the bloodstream, each of which can infect a red blood cell.
- Within the red blood cell, the merozoite develops to form either an erythrocytic stage (blood stage) schizont or a spherical or banana-shaped uninucleate gametocyte.
- The gametocyte, which is the sexual stage of the parasite, is infectious for mosquitoes that ingest it while feeding.
- Within the mosquito, gametocytes develop into female and male gametes (macrogametes and microgametes, respectively), which undergo fertilization and then develop into sporozoites that can infect humans.
- Symptoms of malaria include fever, chills, arthralgia, vomiting, anemia caused by hemolysis, hemoglobinuria, and convulsions.
- There may be the feeling of tingling in the skin, particularly with malaria caused by P falciparum.
- The classical symptom of malaria is a cyclical fever pattern occurring every two days in P. falciparum, P. vivax, and P. ovale infections (tertian fever).
- Severe malaria is almost exclusively caused by P. falciparum infection and is associated with coma and death if untreated.
- Splenomegaly, severe headache, cerebral ischemia, hepatomegaly, and hemoglobinuria with renal failure, i.e., black water fever, may also be seen in severe malaria.
- Young children and pregnant women are especially vulnerable.
- Chronic malaria is seen in both P. vivax and P. ovale infections, but not in P. falciparum, where only the acute fulminating form is usually observed.
- In the chronic form of malaria, the disease can relapse months or years after initial infection, with recurrent waves of parasitemia emanating from the persistence of latent parasites in the liver.
Protective immunity
- Hosts can mount a level of protective immunity, which can occur following initial infection and render the host shielded against subsequent disease.
- Individuals who are repeatedly exposed to malaria develop antibodies against the sporozoite, liver-stage, blood-stage, and/or sexual-stage malaria antigens.
- It is thought that antibodies acting directly against these antigens are responsible for the decreased susceptibility to malaria infection and disease seen in adults in malaria-infested areas.
- Antibodies directed against the sexual stages of plasmodia may also reduce malaria transmission.
- Additional components of naturally acquired immunity include the release of cytokines that act against all stages of the parasite and also a cytotoxic T cell response directed at liver stages of the parasite.
- Below are some of the presumed mechanisms of adaptive immunity to malaria.
- Antibodies block invasion of sporozoites into liver cells.
- IFN-y and CD8 T cells inhibit parasite development in hepatocytes.
- Antibodies block invasion of merozoites into erythrocytes.
- Antibodies prevent sequestration of infected erythrocytes by preventing binding to adhesion molecules on the vascular endothelium.
- IFN-y and CD4+ T cells activate macrophages to phago-cytose intra-erythrocytic parasites and free merozoites.
- Antibodies neutralize parasite glycosylphosphatidylinositol and inhibit induction of the inflammatory cytokine cascade.
- Antibodies mediate complement-mediated cytolysis of extracellular gametes and prevent fertilization of gametes and the development of zygotes. (From Stevenson MM, Riley EM. Innate immunity to malaria. Nat Rev Immunol. 2004;4:169-80)
Quiz
Related Talk
Jeff Dorfman, University of Cape Town – The Immune Mechanisms of Fighting a Malaria Infection
Immunopaedia case study
A 17 year old male with fever and decreased level of consciousness










