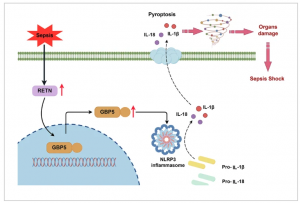
Sepsis remains a major clinical challenge, with limited targeted therapies available. New research identifies the inflammatory molecule resistin (RETN) as a key driver of sepsis progression through its effects on macrophages (Figure 1).
Single-cell analyses showed that RETN is predominantly expressed in monocytes and macrophages, and its levels are significantly elevated in patients with sepsis. Higher RETN expression correlated strongly with increased pro-inflammatory cytokines and greater disease severity.
Mechanistic studies revealed that RETN promotes macrophage pyroptosis, a highly inflammatory form of programmed cell death. Knocking down RETN reduced pyroptosis in vitro and improved survival in septic animal models. Further investigation identified GBP5 and the NLRP3 inflammasome as downstream mediators. RETN overexpression increased GBP5 and NLRP3 activity, whereas RETN suppression reduced inflammasome activation and protected against organ damage in the lungs, spleen, and heart.
Importantly, silencing GBP5 reversed the harmful effects of RETN overexpression, confirming that the RETN-GBP5-NLRP3 pathway is central to macrophage-driven inflammation in sepsis.
These findings define a previously unrecognized signalling axis that amplifies inflammatory cell death during sepsis (Figure 1) and suggest that targeting RETN or its downstream mediators may offer new therapeutic strategies for this life-threatening condition.
Journal article: Chen, Z., et al. 2026. RETN exacerbates sepsis by GBP5/NLRP3 signaling pathway-mediated pyroptosis of macrophage. Gene and Immunity.
Summary by Stefan Botha