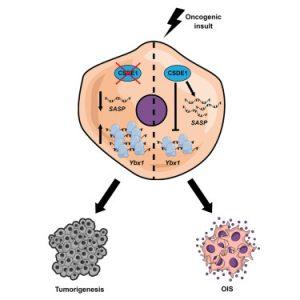
In a recent paper from Cell Reports researchers have highlighted brought to light, new insights into the molecular mechanisms that dictate our body’s natural ability to defend itself against skin cancer (Figure 1). These insights may open the door for improved therapeutics and targets for the treatment of the disease.
Avolio, et al., reported that the protein CSDE1 coordinates a complex chain of events that enable senescence in skin cells, significantly slowing down their function without causing death. The result, cells that acting like a wall to protect one against cancer, further suppressing the formation of tumours.
Through collection of keratinocytes from mice, the most abundant type of skin cell in the epidermis, the researchers were able to investigate their hypothesis. Interestingly, keratinocytes can give rise to various types of skin cancer including basal and squamous cell carcinomas, the most common form of cancers in humans.
Through induction of cell senescence by promoting cancer growth within the cells through manipulation of certain genes, they reported a reduced level of CSDE1. This led to cells becoming immortalised, an essential step in cancer progression.
When CSDE1 was reduced in cells and implanted under the skin of mice, they started forming malignant tumours. Additionally, they discovered that CSDE1 promotes tumour suppression via two different mechanisms (Figure 2). CSDE1 promotes cytokine expression and enzyme production in the cell, forcing a state of growth arrest. It also stops the synthesis of YBX1, a protein known to promote tumour cell proliferation.
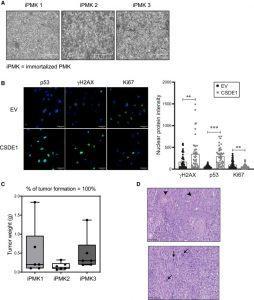
Figure 2: (A) Bright-field microscopy images of three immortalized shCSDE1 cell lines, showing the typical appearance of proliferating cells. Scale bars: 400 μm. (B) Over-expression of CSDE1 in iPMKs decreases proliferation and restores expression of senescence markers. iPMK cells were infected with a tRFP-containing vector expressing or not sh-resistant CSDE1, sorted by fluorescence-activated cell sorting (FACS) at 4 dpi, and tested at 5 dpi for expression of the indicated markers by immunofluorescence using a Stellaris 5 Leica inverted laser scanning confocal microscope. EV, empty vector. Scale bars: 50 μm. Quantification is shown on the right. Significance was assessed by unpaired Student’s t test (∗∗p < 0.01 and ∗∗∗p < 0.001). Error bars represent SEM. (C) iPMK cells are transformed. Cells were injected subcutaneously into nude mice, and tumors were resected and weighted (three mice were injected in two flanks per cell line). Error bars represent SEM. (D) Histopathological analysis of tumors. All tumors were classified as squamous cell carcinomas of grades II to III, with pleomorphic malignant epithelial cells containing large basophilic nuclei (arrows) and focal areas of squamous differentiation (arrowheads). Scale bars: 100 μm (Avolio, et al., 2022).
What was interesting is that previously CSDE1 was linked to driving the formation of cancers and did not suppressing them. CSDE1 is an RNA-binding protein, a type of protein that regulates RNA synthesis and translation.
This is a relatively novel study, investigating RNA-binding proteins and how they establish cell senescence which offers major insights into our understanding of cancer.
Journal article: Avolio, R., et al., 2022. Coordinated post-transcriptional control of oncogene-induced senescence by UNR/CSDE1. Cell Reports.
Summary by Stefan Botha