mTOR inhibitors such as rapamycin are among the most consistent life-extending interventions identified in experimental research. However, how they exert these benefits in humans has remained unclear. A new study suggests that low-dose mTOR inhibition may directly protect the genome, offering a new explanation for its anti-aging effects (Figure 1).
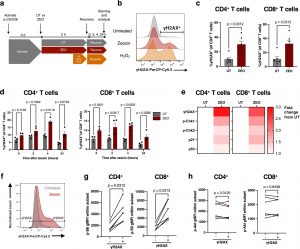
Figure 1: T cell DNA damage is associated with elevated mTORC1 activity. (a) DNA damage assay design. (b) Representative histograms of γH2AX levels by flow cytometry following 4-h recovery in untreated, zeocin-treated (200 μg/mL) and H2O2-treated (25 μM) PBMCs gated on CD4+ T cells. (c, d) Proportion of γH2AX+ of CD4+ (left) and CD8+ (right) T cells after (c) 4 h recovery from zeocin treatment, n = 6 healthy donors or (d) across different recovery times after zeocin treatment, from 4 independent experiments using PBMCs from 1 donor. (e) Heatmaps for levels of DDR signalling molecules expressed as fold change from untreated (UT) cells. (f) Representative gating of γH2AX+ and γH2AX− cells based on untreated (grey), zeocin-treated (red) cells, and fluorescence minus one (FMO, dotted grey) control. (g, h) Geometric mean fluorescence intensity (gMFI) of (g) p-S6 and (h) p-Akt in zeocin-treated CD4+ or CD8+ T cells gated as either positive or negative for γH2AX, n = 6 healthy donors. p-values are derived from a two-way ANOVA with Šídák’s multiple comparisons test (d), and a Wilcoxon matched-pairs signed rank test (c, g, h).
While rapamycin is known to reduce cellular senescence at non-immunosuppressive doses, researchers questioned whether it could also enhance genome stability. Because DNA damage is a key driver of immune aging, the team tested whether mTOR inhibition could reduce genotoxic stress in human T cells.
Rapamycin and related drugs reduced DNA damage and improved cell survival in T cells exposed to acute stress. Notably, this protective effect was not due to reduced protein synthesis, slowed cell division, or increased autophagy, but rather to a direct reduction in DNA lesions.
Analysis of immune cells from older adults showed increased markers of DNA damage, cellular senescence, and overactive mTOR signalling, suggesting that immune aging may be particularly responsive to mTOR modulation. To test this clinically, researchers conducted a placebo-controlled study in older adults receiving low-dose rapamycin. Immune cells from treated participants showed significantly lower levels of p21, a marker associated with DNA damage-induced senescence, compared to placebo.
These findings identify direct genome protection as a previously unrecognized mechanism of mTOR inhibition. This genoprotective effect may help explain rapamycin’s broad geroprotective profile and suggests potential applications in conditions driven by DNA damage, including healthy aging, radiation exposure, and possibly even space travel.
Journal article: Kell. L, et al. 2026. Rapamycin Exerts Its Geroprotective Effects in the Ageing Human Immune System by Enhancing Resilience Against DNA Damage. Aging Cell.
Summary by Stefan Botha










