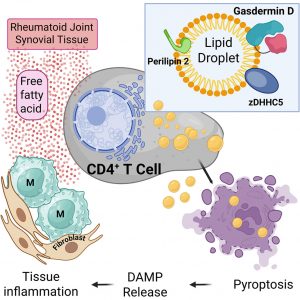
New research provides insight into how metabolic conditions within inflamed joints may worsen inflammation in Rheumatoid arthritis (RA) (Figure 1). The study shows that lipid-rich environments in RA joints can trigger a highly inflammatory form of cell death in immune cells, helping sustain chronic inflammation.
RA is an autoimmune disease in which immune cells infiltrate joint tissue and attack the synovium, leading to pain, swelling, and tissue damage. Among these immune cells, CD4+ T cells play a key role in driving inflammatory responses.
Researchers found that synovial tissue from people with RA contains high levels of free fatty acids (FFAs), creating a lipid-rich microenvironment. These elevated lipid levels were strongly associated with higher inflammation scores in affected joints.
Compared with healthy controls, CD4+ T cells isolated from individuals with RA accumulated more intracellular fatty acids. Lipid droplet analysis showed enrichment of specific fatty acids, including Oleic acid and Myristic acid. These lipid-loaded cells also displayed signs of cellular stress, including endoplasmic reticulum stress and impaired mitochondrial function.
The study revealed that lipid droplets within RA CD4+ T cells contain key proteins involved in inflammatory cell death, including Gasdermin D (GSDMD), Caspase‑1, and Perilipin‑2 (PLIN2).
Exposure to fatty acids triggered rapid cell lysis in CD4+ T cells from RA patients through Pyroptosis, a form of programmed inflammatory cell death. During this process, lipid droplets helped transport gasdermin D to the plasma membrane, where it formed pores that caused the cell to rupture and release inflammatory molecules into surrounding tissue.
Importantly, this lipid-induced pyroptosis occurred in CD4+ T cells from RA patients but not in cells from healthy controls, suggesting that autoimmune T cells are uniquely vulnerable to metabolic stress.
The researchers also tested whether disrupting this pathway could reduce inflammation. In a mouse model of RA, two strategies proved effective:
- Blocking acylation of gasdermin D, which prevents its activation
- Reducing lipid droplet formation by knocking down perilipin-2
Both interventions reduced the recruitment of CD4+ T cells to joints and significantly alleviated synovial inflammation.
Together, the findings suggest that CD4+ T cells in RA are metabolically primed to undergo pyroptosis within the lipid-rich synovial environment. When these cells rupture, they release inflammatory molecules that further amplify joint inflammation.
The study highlights how metabolic stress and immune signaling intersect in autoimmune disease. Targeting lipid metabolism or pyroptosis pathways may therefore represent a promising strategy for reducing inflammation and slowing disease progression in rheumatoid arthritis.
Journal article: Kumar, J., et al. 2026. Lipid droplet-induced T cell death sustains autoimmune tissue inflammation. Cell Metabolism.