Dexamethasone is a corticosteroid that was put into clinical trial to identify whether it could modulate immune-mediated lung injury and reduce progression to respiratory failure and death in COVID-19 patients. In a pre-peer reviewed paper, preliminary results of the RECOVERY trial was reported. This was a randomized, controlled, open-label, adaptive, platform trial comparing a range of possible treatments with usual care in patients hospitalized with COVID-19. 2104 patients randomly received dexamethasone, 6 mg given once daily for up to ten days, and compared with 4321 patients provided with usual care. The primary outcome of the trial was mortality at 28 days and overall dexamethasone treated patients had a 21.6% mortality compared with 24.6% for patients with usual care and not receiving drug. The authors state that “Dexamethasone reduced deaths by one-third in patients receiving invasive mechanical ventilation…… by one-fifth in patients receiving oxygen without invasive mechanical ventilation …… but did not reduce mortality in patients not receiving respiratory support at randomization”. It was concluded that Dexamethasone, being commonly available worldwide, can provide effective treatment for patients with severe COVID-19.
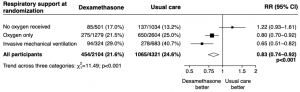
Effect of allocation to dexamethasone on 28−day mortality by level of respiratory support received at randomization. How effective is Dexamethasone for the treatment of patients with COVID-19? (Source: Horby et al., Pre-print)
Article: Horby et al., Pre-Print. Effect of Dexamethasone in Hospitalized Patients with COVID-19: Preliminary Report. MedRviv
Summary by Clive Gray










