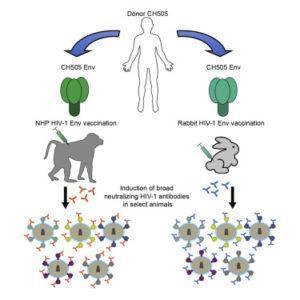
One goal in HIV vaccine research is the development of an antibody-based vaccine to prevent HIV acquisition. Eliciting antibodies which can prevent entry of various strains of HIV has proven challenging. Researchers from Duke University have now tested Envelope (Env) immunogens in rabbits and macaques and found that they can elicit broadly neutralizing antibody (bnAb) responses. This study highlights the potential for Env immunogens as HIV vaccines.
BnAbs are of particular interest in the HIV vaccine field as they can neutralize a wide range of the diverse HIV strains in circulation. Multiple immunogens have been designed to elicit broad antibody responses, however there has been little success in this area. As the sole target of bnAbs is the HIV Env, immunogens based on this protein may produce the required broad response.
Researchers, led by Barton Haynes, investigated whether optimized immunogens based on the Envs of transmitted/founder viruses would be able to produce bnAb response in animal models as a proof of concept study. The team designed the immunogens to elicit antibodies towards the CD4-binding site, a key target of bnAbs. They immunized both rabbits and macaques.
Upon immunization, some animals produced bnAbs which were able to neutralize diverse, moderately antibody-resistant HIV strains. Interestingly, the antibodies which were elicited did not target the CD4-binding site but targeted another bnAb epitope at the top of the Env trimer, the V1/V2-glycan site. These data suggest that V1/V2-glycan site-specific bnAbs may be more easily produced than CD4-binding site-specific antibodies upon immunization.
Unfortunately, most of the animals did not develop bnAbs upon immunization, which further highlights the difficulty of producing such antibodies with the current Env immunogens available. The use of a variety of Env immunogens, administered sequentially could result in more broad responses and this will be tested in the future.
In summary, this study highlights that bnAb responses can be elicited in animal models and this may translate to human studies. Furthermore, the data also suggest that eliciting V1/V2-glycan site-specific antibodies may be easier than eliciting those towards the CD4-binding site. Vaccine efforts may need to shift to focus more on the V1/V2-glycan epitope.
Journal Article: Saunders et al. 2017. Vaccine Induction of Heterologous Tier 2 HIV-1 Neutralizing Antibodies in Animal Models. Cell Reports
Journal Article by Thandeka Moyo