A new gene therapy has received approval from the U.S. Food and Drug Administration, marking the first approved treatment for the rare genetic immune disorder Leukocyte Adhesion Deficiency Type I (LAD-I).
The therapy, developed through decades of research led by Donald Kohn at UCLA, provides a potentially life-saving option for children affected by this severe condition.
LAD-I is an extremely rare paediatric disorder, affecting roughly one in a million children worldwide. It is caused by mutations in the ITGB2 gene, which normally produces proteins that allow white blood cells to travel through the bloodstream and reach sites of infection.
When this mechanism fails, the immune system cannot properly respond to bacteria or fungi. As a result, children experience recurrent and severe infections, and without effective treatment, survival beyond childhood is uncommon.
The new treatment, marketed as Kresladi, uses an advanced gene therapy approach. Doctors collect a child’s own blood stem cells, insert a healthy copy of the defective gene, and then return the modified cells to the patient.
Once inside the body, these corrected cells can generate functioning immune cells capable of fighting infections and healing wounds. Because the therapy uses the patient’s own cells, it avoids complications often associated with donor transplants, such as immune rejection or graft-versus-host disease.
The approval was based on results from a clinical trial conducted across several international sites. Nine children between five months and nine years old participated in the study.
The results were highly encouraging (Figure 1):
- All nine patients survived without requiring bone marrow transplantation.
- No cases of immune rejection or graft failure occurred.
- Severe infections requiring hospitalization dropped significantly.
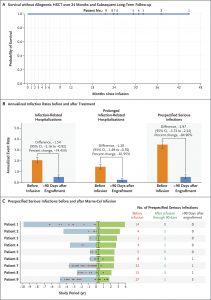
Figure 1: Hematopoietic Stem-Cell Transplantation (HSCT)–free Survival and Reduction in Incidence of Infections.
Researchers also observed sustained expression of the restored immune proteins and improved immune function in treated children.
It is the first therapy from his research program to achieve regulatory approval, making it possible for patients to access the treatment through specialized medical centres.
The work was supported in part by the California Institute for Regenerative Medicine, which funds stem cell and gene therapy research.
While the therapy has shown remarkable promise, long-term monitoring of treated patients will continue through clinical follow-up studies and registries to confirm its sustained benefits.
If similar breakthroughs follow, gene therapy could transform how many rare childhood diseases are treated, turning once-fatal conditions into manageable or even curable disorders.
Clinical trial: Kohn, D., et al. 2026. Long-Term Follow-Up (LTFU) for Gene Therapy of Leukocyte Adhesion Deficiency-I (LAD-I). Clinicaltrials.gov
Manuscript: Booth, C., et al. 2025. Lentiviral Gene Therapy for Severe Leukocyte Adhesion Deficiency Type 1. NEJM.
Summary by Stefan Botha










