Researchers are now focusing on sepsis in a new study after discovering a unique mechanism for platelet activation in pathogenic blood clotting/thrombosis (Figure 1).
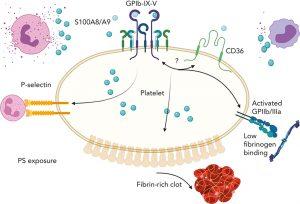
The anti-microbial protein S100A8/A9, which is secreted from activated immune cells, and the platelet receptor glycoprotein I alpha (GPIb?) have been shown to form a hitherto unidentified axis. The process is different from the well-known “clotting cascade” that prevents excessive blood loss after damage and is not stopped by the traditional anti-platelet medications now used to treat arterial thrombosis.
There are two primary populations of platelets that are present when clots develop at the site of a vascular lesion. Procoagulant platelets are situated at the shell around the centre of the clot and aid in the production of fibrin, which stabilises the clot, whereas highly active and aggregated platelets are found at the clot’s core.
Although it does not cause platelet aggregation, the S100A8/A9-GPIb? axis does cause the creation of procoagulant platelets and speeds up fibrin activation and thrombosis. This axis may be especially important in diseases when platelet and innate immune cell activation are crucial for clotting.
The interaction of S100A8/A9 with various receptors makes it an intriguing target to reduce both clotting and inflammation during sepsis, according to the researchers, who believe that this novel mechanism may be relevant in thrombosis observed under chronic inflammatory conditions and acute infections.
Journal article: Colicchia, M., et al., 2022. S100A8/A9 drives the formation of procoagulant platelets through GPIbα. Blood.
Summary by Stefan Botha