Inflammatory bowel disease (IBD) remains one of the most challenging chronic inflammatory disorders to treat. Despite advances in biologic therapies, many patients with conditions like Ulcerative colitis and Crohn disease continue to experience persistent inflammation, tissue damage, and treatment resistance.
Part of the challenge lies in the complexity of the disease itself. IBD is not driven by a single immune pathway or cell type, it is a constantly evolving interaction between immune cells, intestinal epithelium, stromal tissue, and microbial signals.
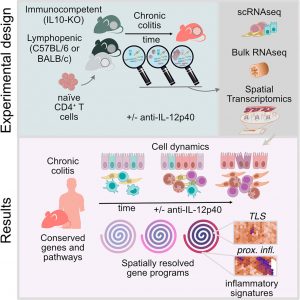
A new study takes a major step toward understanding this complexity by creating a high-resolution, longitudinal atlas of chronic colitis using bulk transcriptomics, single-cell RNA sequencing, and spatial transcriptomics across multiple disease models (Figure 1).
The result is a dynamic view of how inflammation evolves over time, and which cellular programs remain conserved across both mice and humans.
Using two well-established murine models of colitis, T cell transfer colitis and spontaneous Il10-deficient colitis, researchers tracked disease progression over time and integrated molecular data at multiple levels.
Despite their different triggers, both models converged on remarkably similar inflammatory states. Across datasets, one cell population repeatedly emerged as a dominant driver of chronic disease: Neutrophils.
Neutrophils expanded dramatically during disease progression and were linked to inflammatory pathways that strongly resembled those observed in human ulcerative colitis. Cytokine signalling and neutrophil-associated transcriptional programs proved to be among the most conserved features across species.
This finding reinforces the growing idea that neutrophils are not simply short-lived inflammatory responders, but major orchestrators of chronic intestinal pathology.
Another striking observation was the progressive transformation of intestinal epithelial cells during chronic inflammation.
As disease advanced, epithelial cells increasingly upregulated antigen presentation pathways, suggesting they actively participate in immune activation rather than merely responding to injury. This shift may fundamentally alter how epithelial cells interact with T cells and other immune populations within the gut.
The findings support a newer view of the epithelium as an active immunological interface, one capable of amplifying and sustaining inflammation when chronically stressed.
One of the most powerful aspects of the study was its use of spatial transcriptomics, allowing researchers to map where specific inflammatory programs emerge within tissue architecture.
Rather than being uniformly inflamed, diseased tissue contained highly organized inflammatory domains. Among these were structures resembling Tertiary lymphoid structures, localized immune hubs that form during chronic inflammation and may support ongoing immune activation.
Distinct epithelial and immune programs occupied separate spatial regions, highlighting that chronic colitis is not a diffuse process but a patchwork of specialized inflammatory niches.
This spatial organization may help explain why some therapies only partially work: different inflammatory microenvironments within the same tissue may respond differently to treatment.
No single mouse model fully captures human IBD. By integrating multiple complementary systems, the researchers were able to reconstruct a broader and more realistic picture of disease progression.
Some models better captured adaptive immune activation, while others emphasized spontaneous innate inflammation or tissue remodelling. Together, they recreated many of the cellular and transcriptional features seen in human disease.
This integrated approach provides a framework for future translational studies aimed at identifying which pathways are truly conserved, and therefore most therapeutically relevant.
Beyond its biological findings, the study delivers something equally valuable: a detailed spatiotemporal atlas of chronic intestinal inflammation.
This resource may help researchers:
- Track how inflammatory programs evolve over time
- Identify early versus late disease mechanisms
- Understand why therapies fail in some patients
- Discover spatially restricted therapeutic targets
Most importantly, it moves the field closer to a systems-level understanding of IBD, where disease is viewed not as a single pathway gone wrong, but as a coordinated and evolving ecosystem of immune, epithelial, and stromal interactions.
Journal article: Fransson, J., et al. 2026. Spatiotemporal analysis reveals distinct inflammatory programs underlying chronic colitis. Immunity.
Summary by Stefan Botha