Antibody-based therapies have transformed modern medicine, offering highly targeted treatments for cancer, autoimmune diseases, and chronic inflammation. But in rare cases, these powerful biologics can trigger a dangerous immune overreaction, anaphylaxis, a rapid and potentially fatal allergic response. Until now, predicting which antibody drugs carry this risk has remained a major challenge.
A new study sheds light on a critical mechanism that may help explain, and eventually prevent, these severe reactions (Figure 1).
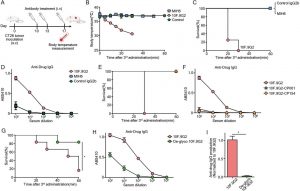
Figure 1: High-affinity FcγR-binding anti-PD-L1 antibody clone, 10F.9G2, but not low-affinity clones nor deglycosylated 10F.9G2 induced anaphylaxis. (A) Experimental scheme for treatment of CT26 tumor-bearing mice with anti-mouse PD-L1 antibodies. CT26 tumor-bearing mice were treated with antibodies (200 µg/mouse) on days 10, 13 and 17 after inoculation. Body temperature and survival were measured after the third antibody injection. (B) Changes in body temperature of CT26 tumor-bearing mice after the third injection of 10F.9G2 (n=4), Control IgG2b (n=4), or MIH6 (n=3). (C) Survival curve of CT26 tumor-bearing mice after the third injection of antibodies in (B). (D) Concentrations of IgG ADA against 10F.9G2 (n=6), Control IgG2b (n=6), and MIH6 (n=4) in serum evaluated by ELISA. (E) Survival of CT26 tumor-bearing mice after the third injection of 10F.9G2 (n=4), 10F.9G2-CP001 (n=4), or 10F.9G2-CP154 (n=4). (F) Concentrations of IgG ADA against 10F.9G2 (n=4), 10F.9G2-CP001 (n=4), and 10F.9G2-CP154 (n=4) in the serum evaluated by ELISA. (G) Survival of CT26 tumor-bearing mice after the third injection of 10F.9G2 (n=6) or deglycosylated 10F.9G2 (n=6). *p<0.05, using log-rank (Mantel-Cox) tests. (H) Serum concentrations of the IgG ADAs against 10F.9G2 (n=3) and deglycosylated 10F.9G2 (n=3) evaluated by ELISA. (I) Normalized ADA production against 10F.9G2 (n=3) and deglycosylated 10F.9G2 (n=3) in serum. *p<0.05, unpaired t-test with Welch’s correction. Data are represented as mean±SE. ADAs, antidrug antibodies; FcγR, Fcγ receptor; i.v., intravenous; PD-L1, programmed death-ligand 1; s.c., subcutaneous.
Traditionally, anaphylaxis has been understood through the lens of IgE-mediated immunity: allergens trigger B cells to produce IgE antibodies, which activate mast cells and basophils to release histamine. However, this framework doesn’t fully explain reactions seen with some therapeutic antibodies.
This study points to an alternative pathway, one driven by Fc gamma receptors (FcγRs), immune receptors that bind to the Fc (constant) region of antibodies and regulate how immune cells respond to them. The researchers discovered that antibody drugs with higher affinity for FcγRs are more likely to trigger harmful immune responses. In particular:
- Strong FcγR binding increased the likelihood that the immune system would recognize the therapeutic antibody as “foreign”
- This led to elevated production of anti-drug antibodies (ADAs)
- High ADA levels were closely associated with severe, rapid-onset anaphylaxis
In mouse models, one anti–PD-L1 antibody with strong FcγR binding triggered fatal anaphylaxis in all treated animals, while a similar antibody with weaker binding caused minimal immune activation and no severe reactions.
A critical piece of the puzzle lies in tumour-associated myeloid cells. These immune cells were found to:
- Efficiently capture antibodies with strong FcγR binding
- Process and present them in a way that amplifies immune activation
- Drive robust ADA production
When FcγRs were experimentally blocked, this cascade was disrupted, reducing ADA levels and preventing anaphylaxis. This strongly suggests that FcγR-mediated uptake is a central trigger in this pathway.
These findings have immediate relevance for drug development. If FcγR binding strength influences immunogenicity and safety, then engineering antibodies with optimized (or reduced) FcγR interactions could:
- Lower the risk of ADA formation
- Reduce the likelihood of severe allergic reactions
- Improve the overall safety profile of antibody therapeutics
Rather than abandoning highly potent antibodies, the goal becomes fine-tuning immune engagement.
As antibody therapeutics continue to expand across medicine, integrating immunological precision into their design will be essential. Fcγ receptors, once considered secondary players, may now be central to that effort.
Journal article: Tang, R., et al. 2026. Antibody therapeutics with high affinity for FcγRs exacerbate anaphylaxis via FcγR-mediated capture by tumor- associated myeloid cells. Journal of ImmunoTherapy for Cancer.
Summary by Stefan Botha










