Systemic lupus erythematosus (SLE) is typically defined by dysfunctional B cell responses, autoantibodies and immune complex deposition are considered its central drivers. But this narrative has always had a gap: the strongest predictor of poor outcomes in lupus nephritis isn’t antibodies, it’s T cell infiltration in the kidney.
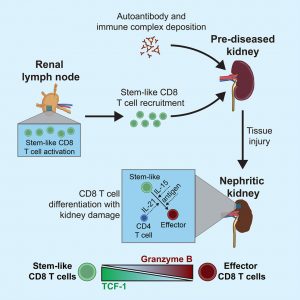
A new study reframes this paradox, revealing that CD8-positive T cells are not just bystanders in lupus nephritis, they are active drivers of tissue damage, following a highly organized and antigen-driven developmental program (Figure 1).
The story begins not in the kidney, but in the renal-draining lymph nodes. Here, a population of stem-like CD8⁺ T cells, marked by TCF-1 expression, acts as a reservoir.
These cells:
- Undergo T cell receptor (TCR)-dependent activation
- Expand clonally in response to antigen
- Maintain a “stem-like” state that allows continued proliferation and differentiation
Rather than being terminally differentiated from the outset, these cells behave more like a renewable source of pathogenic T cells.
Once these stem-like cells migrate into the kidney, they undergo a critical shift.
Within the tissue, they:
- Differentiate into highly cytotoxic effector cells
- Acquire the ability to directly damage renal tissue
- Expand further in an antigen-dependent manner
This local differentiation step is key—it’s not just the presence of T cells, but their activation state within the kidney microenvironment that determines pathology.
Two cytokines emerge as central regulators of this process:
- IL-15: supports survival and activation of CD8⁺ T cells
- IL-21: enhances differentiation and effector function, often linked to CD4⁺ T cell help
Together, these signals drive the transition from stem-like precursors to fully armed cytotoxic T cells. Notably, this process also depends on help from CD4-positive T cells, highlighting coordinated immune crosstalk.
In chronic infections and cancer, prolonged antigen exposure often leads to T cell exhaustion, where cells lose their effector capacity despite expressing inhibitory receptors.
Here’s the twist: in lupus nephritis, CD8⁺ T cells show features of chronic activation, such as immune checkpoint expression, but retain strong cytotoxic function.
This means:
- They are not functionally exhausted
- They continue to produce inflammatory mediators
- They sustain tissue damage over time
Persistent activity of transcription factors like AP-1 reinforces this pathogenic state.
Interestingly, the differentiation trajectory observed in lupus mirrors programs seen in:
- Chronic viral infections
- Tumour-infiltrating lymphocytes in cancer
However, the outcome diverges. Instead of becoming dysfunctional, these T cells remain fully capable killers, turning their activity against host tissue.
Importantly, similar CD8⁺ T cell programs were observed in human lupus nephritis, suggesting this is not just a mouse model artifact but a conserved disease mechanism.
This work challenges the traditional B cell-centric view of SLE by placing CD8⁺ T cells at the center of organ damage in lupus nephritis.
It suggests that:
- Targeting T cell differentiation pathways may be as important as modulating antibodies
- Interrupting signals like IL-15 or IL-21 could limit tissue-specific damage
- Preventing the transition from stem-like to cytotoxic states may offer a new therapeutic angle
In essence, lupus nephritis may not just be an antibody-driven disease, it’s also a T cell-driven tissue attack, orchestrated with precision and sustained by chronic immune activation.
Journal article: Al Souz, J., et al. 2026. Organ injury in systemic autoimmunity is mediated by stem-like CD8+ T cells arising from tissue-draining lymph nodes. Immunity.
Summary by Stefan Botha