Glucagon-like peptide-1 receptor agonists (GLP-1RAs) have become a cornerstone in the treatment of type 2 diabetes, widely prescribed for their ability to improve glycaemic control and support weight loss. But as their use expands, so does interest in their broader immunological effects. A new study takes a deep dive into this question, uncovering genetic evidence that these therapies may influence susceptibility to certain autoimmune diseases, particularly those affecting the gut (Figure 1).
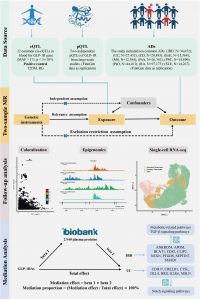
To investigate this, researchers applied a multi-omics Mendelian randomization framework, integrating genomic, transcriptomic, epigenomic, and proteomic data. By using genetic variants that regulate the GLP-1 receptor as proxies for drug exposure, they were able to estimate the long-term biological effects of GLP-1RA activity.
The findings point to a specific and somewhat unexpected association. Increased GLP-1 receptor activity was linked to a higher risk of ulcerative colitis, a chronic inflammatory condition of the colon. Notably, this association did not extend to other autoimmune diseases or even to Crohn’s disease, suggesting a degree of disease specificity rather than a broad immune effect.
Digging deeper, the study identified a key genetic variant, rs9283907, that appears to regulate GLP-1 receptor expression and is also associated with ulcerative colitis risk. This overlap suggests a shared biological mechanism linking metabolic signalling to intestinal inflammation. Supporting this, the researchers found that GLP-1 receptor expression is altered in colon tissue from patients with ulcerative colitis, with particularly strong enrichment in intestinal stem cells. These cells are essential for maintaining and repairing the gut lining, placing them at a critical junction between tissue integrity and immune activation.
The mechanistic insights become even more compelling when considering downstream signalling pathways. The study highlights the involvement of the Notch signalling pathway, mediated in part by proteins such as DLL4. This pathway is known to regulate cell fate decisions and immune responses in the gut, and its modulation could influence how the intestinal barrier responds to injury or inflammation. Mediation analysis suggests that changes in this signalling axis may explain a substantial portion of the increased ulcerative colitis risk associated with GLP-1 receptor activity.
Taken together, these findings offer the first genetic evidence that GLP-1–targeting therapies may have unintended effects on intestinal immune homeostasis. While this does not diminish their clinical value, particularly given their well-established benefits, it does highlight the importance of understanding how metabolic interventions intersect with immune regulation.
Journal article: Li, H., et al. 2026. Integrative multi-omics analysis reveals GLP-1R agonists-driven proteins via Notch signalling as mediators of ulcerative colitis risk. Genes & Diseases.
Summary by Stefan Botha