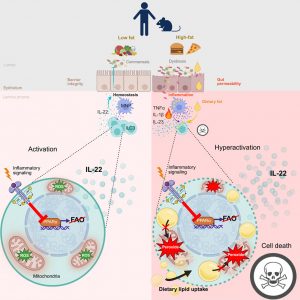
The gut is more than a digestive organ, it is a highly responsive immune environment, constantly integrating signals from diet, microbes, and host tissues. A recent study highlights just how rapidly this balance can be disrupted, revealing that high-fat diets can directly impair key immune cells in the intestine (Figure 1).
At the centre of this discovery are group 3 innate lymphoid cells (ILC3s), a population of immune cells that act as environmental sentinels in the gut. These cells play a critical role in maintaining barrier integrity, coordinating responses to microbes, and preserving intestinal homeostasis. Because they sit at the interface between diet and the microbiota, ILC3s are uniquely positioned to respond to changes in the gut environment, but this also makes them vulnerable.
The study shows that exposure to a high-fat diet leads to a rapid and marked loss of intestinal ILC3s in both mice and humans. Strikingly, this effect occurs within hours and is independent of body weight, caloric intake, or glucose metabolism. Instead, the trigger appears to be an immediate shift in the gut environment. High-fat intake alters the microbiota and increases intestinal permeability, allowing microbial signals to activate inflammatory mononuclear phagocytes. This creates an inflammatory milieu that profoundly affects ILC3 function.
What follows is a breakdown in cellular metabolism. Inflammatory signalling suppresses fatty acid oxidation, a pathway that ILC3s rely on to process lipids and sustain their energy needs. As fatty acids accumulate, the cells experience mitochondrial stress, leading to dysfunction and ultimately cell death, a process described as lipotoxicity. Notably, this vulnerability appears to be highly specific: other immune cells in the same environment, such as TH17 cells, do not exhibit the same metabolic collapse.
This selective sensitivity points to a deeper principle in immunology. Immune cells are not metabolically interchangeable; they are finely tuned to energy pathways that support their function. For ILC3s, this specialization becomes a liability in the context of a lipid-rich, inflammatory environment. Importantly, the study demonstrates that this mechanism is conserved across species, strengthening its relevance to human health.
Encouragingly, the damage is not permanent. When excess dietary fat is removed, ILC3 populations can recover, suggesting that this immune dysfunction is reversible and tightly linked to environmental conditions. This opens the door to potential interventions that leverage diet to restore immune balance.
Taken together, these findings reshape how we think about the relationship between nutrition and immunity. Rather than being a downstream consequence of metabolic disease, immune dysfunction in the gut may be an early and direct response to dietary composition. By uncovering how dietary fats and microbial signals converge to drive immune cell loss, this work highlights a new layer of regulation in intestinal health and suggests that restoring metabolic balance may be key to preserving immune resilience.
Journal article: Torrico, E.C., et al. 2026. High-fat diet causes rapid loss of intestinal group 3 innate lymphoid cells through microbiota-driven inflammation and mitochondrial stress. Immunity.
Summary by Stefan Botha