Idiopathic pulmonary fibrosis (IPF) remains one of the most challenging lung diseases to treat, marked by progressive scarring that gradually impairs breathing. While immune cells have long been implicated in this process, new research is sharpening the focus on one key player: macrophages, and how they drive fibrosis at the molecular level.
Using single-cell and bulk transcriptomic datasets, researchers mapped how immune cells behave across healthy lungs, COPD, and IPF (Figure 1). Important findings emerged:
- Monocyte-derived macrophages (MDMs) were significantly increased in IPF
- Higher MDM levels were linked to worse lung function
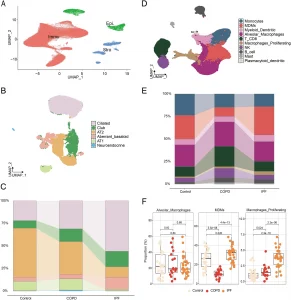
Figure 1: Single cell transcriptome analysis of epithelial cells and immune cells in IPF. (A) UMAP of all single-cell transcriptomes clustered by main cell type; (B) UMAPs of epithelial single-cell transcriptomes color-coded by cell subtypes; (C) Quantification of epithelial cell subtypes per tissue type; (D) UMAPs of all immune single-cell transcriptomes color-coded by cell subtypes; (E) Quantification of epithelial cell types per tissue type; (F) Statistics of macrophage proportions in per tissue type.
This reinforces the idea that not all macrophages are equal, their origin and activation state matter in disease progression.
The study highlights intensified communication between macrophages and structural lung cells:
- Increased interactions between macrophages and fibroblasts / myofibroblasts
- Amplified signalling through the TGF-β signalling pathway, a central driver of fibrosis
This signalling cascade promotes excessive extracellular matrix (ECM) production, the hallmark of fibrotic tissue buildup that stiffens the lungs.
In essence, macrophages are not just bystanders, they actively orchestrate fibrotic remodelling.
To translate these findings clinically, the researchers developed a macrophage-associated gene signature:
- Built using multivariate Cox regression
- Validated across independent datasets
- Demonstrated strong prognostic performance (via ROC analysis)
This signature could help clinicians stratify patients, predict disease progression, and tailor management strategies, moving toward more personalized care in IPF.
Among the identified genes, LGMN (legumain) emerged as a key regulator:
- Highly expressed in M2 macrophages (pro-fibrotic phenotype)
- Co-localized with CD206 in fibrotic lung tissue
- Promoted macrophage–fibroblast communication via TGF-β1 secretion
Crucially, inhibiting LGMN with RR-11a led to:
- Reduced TGF-β1 production
- Disrupted harmful cell–cell signalling
- Attenuation of lung fibrosis in mouse models
This positions LGMN as a promising therapeutic target for interrupting the fibrotic feedback loop.
This study advances IPF research on multiple fronts:
- Mechanistic insight: Identifies how macrophages actively drive fibrosis through intercellular signalling
- Biomarker development: Introduces a robust gene signature for prognosis
- Therapeutic potential: Highlights LGMN inhibition as a strategy to blunt disease progression
Fibrotic diseases like IPF are increasingly understood as disorders of dysregulated immune–stromal communication. This work underscores how targeting cellular cross-talk, not just individual cell types, may be key to effective treatment.
As precision medicine continues to evolve, integrating immune profiling with functional biology could reshape how we diagnose and treat chronic lung diseases.
Journal article: Xia, Y, et al. 2026. LGMN promotes crosstalk between macrophages and fibroblasts in pulmonary fibrosis: a potential therapeutic target. Frontier Immunology.
Summary by Stefan Botha










