Alzheimer’s disease is a progressive neurological disorder that gradually affects memory, thinking, and behaviour. As populations age worldwide, the condition has become the leading cause of dementia and a major public health challenge.
Early diagnosis is critical for managing symptoms and slowing disease progression. However, diagnosing Alzheimer’s currently relies heavily on cognitive testing, brain imaging, and sometimes the measurement of proteins such as Amyloid‑β and Phosphorylated Tau in cerebrospinal fluid. These methods can be expensive, invasive, or difficult to access in many healthcare settings.
Scientists are therefore searching for simpler blood-based biomarkers that could help detect the disease earlier and more widely.
Researchers are exploring new ways to diagnose Alzheimer’s disease earlier and more accurately. A recent study suggests that a blood-based inflammatory marker could play an important role in identifying people at risk of the disease, particularly those with certain genetic risk factors (Figure 1).
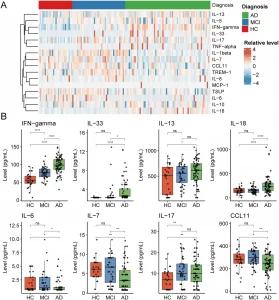
Figure 1: Differential expression of plasma inflammatory proteins among AD, MCI, and HC groups. (A) Heatmap showing the relative expression levels of 16 plasma inflammatory markers in individuals with Alzheimer’s disease (AD), mild cognitive impairment (MCI), and healthy controls (HC). (B) Box plots comparing the plasma levels of eight significantly differentially expressed inflammatory markers among the AD, MCI, and HC groups, including IFN-γ, IL-33, IL-13, IL-18, IL-6, IL-7, IL-17, and CCL11. ns, not significant; *P < 0.05; **P < 0.01; ****P < 0.0001.
A key focus of Alzheimer’s research is the role of neuroinflammation and the brain’s immune cells, known as Microglia. These cells normally help protect the brain by clearing harmful debris, including protein aggregates. But prolonged activation of microglia can release inflammatory molecules that damage neurons and worsen neurodegeneration.
Genetics also plays an important role. The APOE ε4 allele is the strongest known genetic risk factor for late-onset Alzheimer’s disease. People carrying this variant have a significantly higher risk of developing the condition.
Previous studies suggest that APOE ε4 can alter microglial behaviour, promoting chronic inflammation and contributing to disease progression.
To investigate potential diagnostic markers, researchers analysed data from including with Alzheimer’s disease, mild cognitive impairment, and healthy controls.
Participants underwent cognitive testing. Brain scans were also performed to rule out unrelated neurological conditions. Blood samples were collected to measure 16 inflammatory biomarkers, and genetic testing was used to determine each participant’s APOE genotype.
Among the biomarkers examined, the immune signalling molecule Interferon‑gamma (IFN‑γ) emerged as the most powerful indicator of Alzheimer’s disease.
Researchers found that:
- IFN-γ levels were significantly higher in Alzheimer’s patients than in healthy individuals.
- Higher IFN-γ levels were associated with poorer cognitive performance.
- IFN-γ alone could distinguish Alzheimer’s patients from healthy controls with high accuracy.
When combined with clinical information and APOE genotype, a predictive model achieved very strong diagnostic performance.
Further analysis of brain transcriptomic data revealed that inflammatory pathways associated with IFN-γ were particularly active in microglia from patients with the APOE ε4 genotype.
The researchers also identified a subtype of microglia known as lipid-droplet-accumulating microglia, which showed strong activation of IFN-γ–related pathways. Laboratory experiments suggested that APOE ε4 and IFN-γ together may promote these harmful microglial changes.
The study highlights IFN-γ as a promising blood biomarker for Alzheimer’s disease, particularly in people carrying the APOE ε4 genetic variant. These findings suggest that combining blood biomarkers with genetic information could significantly improve early detection.
Journal article: Huang, R., et al 2026. Identification of plasma inflammatory biomarkers for Alzheimer’s disease reveals IFN-γ as a regulator of ACSL1-mediated microglia phenotype. Frontiers in Immunology.
Summary by Stefan Botha










