Human papillomavirus (HPV) is the most common sexually transmitted infection worldwide, and persistent infection with high-risk strains remains the leading cause of cervical cancer. While many HPV infections resolve naturally, understanding how the immune system controls or clears the virus is essential for improving prevention, diagnosis, and treatment strategies.
A recent study investigated how T cells, key players in the immune system, respond to HPV in both vaccinated women and women diagnosed with early cervical lesions known as cervical intraepithelial neoplasia grade 1 (CIN1) (Figure 1). These lesions represent an early stage of abnormal cervical cell growth that can sometimes progress to cancer if persistent infection occurs.
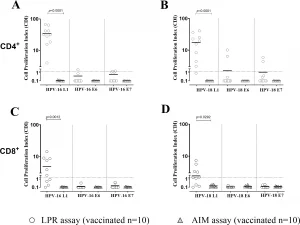
Figure 1: Cell proliferation index (CPI) for both CD4+(A, B) and CD8+ T cells (C, D) against HPV-16 and HPV-18 for antigens L1, E6 and E7 was measured using Lymphoproliferation (LPR; white dots) and Activation Induced Markers (AIM; grey triangles) assays in 18 HPV vaccinated healthy women. Significant p value were given for each graph. Dotted horizontal lines indicates cut-off of positive results.
Researchers evaluated immune responses to proteins from HPV-16 and HPV-18, two high-risk strains responsible for most HPV-related cancers. Using advanced flow cytometry techniques, the team measured how T cells reacted to viral proteins called L1, E6, and E7. Two laboratory approaches were compared: a short-term activation-induced marker (AIM) assay and a longer seven-day lymphoproliferation (LPR) assay.
The findings revealed that the LPR assay was more sensitive in detecting HPV-specific immune responses. Across participants, T-cell responses were primarily directed against the viral L1 protein and were significantly stronger in CD4⁺ T cells than in CD8⁺ T cells.
Among vaccinated women, all participants showed a clear CD4⁺ T-cell response to the HPV-16 L1 protein, with slightly weaker responses against HPV-18. Interestingly, nearly half of the women with CIN1 also displayed HPV-16-specific immune responses, and about one-third showed responses to HPV-18, even when they were infected with different HPV strains. This suggests that immune responses may sometimes recognize shared features across multiple HPV genotypes, a phenomenon known as cross-reactivity.
Further analysis showed that these immune responses were largely associated with TH1 and TH17 immune profiles, both of which are involved in antiviral defence and inflammatory immune signalling.
These findings provide valuable insight into how the immune system interacts with HPV during early stages of infection and disease. By improving methods to detect HPV-specific T-cell responses, researchers hope to better understand why some infections clear naturally while others persist and progress to cancer.
In the future, immune-based testing approaches like these could potentially help clinicians identify which cervical lesions are more likely to progress and which may resolve on their own. Such advances could support more personalised patient monitoring and reduce unnecessary interventions while ensuring that high-risk cases receive timely treatment.
Journal article: Zelini, P., et al. 2026. Detection of human papillomavirus (HPV)-specific T cell response in women with low-grade cervical intraepithelial lesion and HPV vaccinated subjects. Frontiers in Immunology
Summary by Stefan Botha










