Researchers have identified a mechanism that allows certain HPV-associated head and neck cancers to evade immune detection (Figure 1). The study found that these tumours exploit a protein called MARCHF8 to degrade major histocompatibility complex class I (MHC-I) molecules, which normally act as signals that alert the immune system to abnormal cells. Without these markers, cancer cells remain largely invisible to immune surveillance.
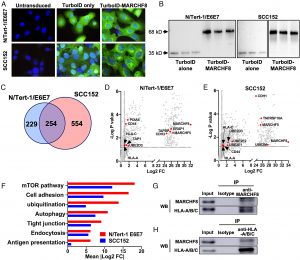
Figure 1: MARCHF8 protein interacts with MHC-I proteins. MARCHF8-TurboID fusion proteins or TurboID proteins alone were stably expressed in N/Tert-1 cells expressing HPV16 oncoproteins E6 and E7 (N/Tert-1/E6E7) and HPV+ HNC cells (SCC152). The TurboID biotinylates the proteins within close proximity. The biotinylated proteins in the cells were separated using streptavidin-coated beads and analyzed using mass spectrometry. Biotinylation by TurboID or TurboID-MARCHF8 was validated by fluorescence microscopy with streptavidin-488 (green) and Hoechst dye (blue) to label DNA (A). Expression of TurboID or TurboID-MARCHF8 was validated by western blot with anti-HA (B). (C) A total of 1,037 proteins enriched with more than a twofold increase and P-value less than 0.05 with MARCHF8-TurboID compared to TurboID control in both N/Tert-1 E6E7 (D) and SCC152 cells (E) were defined as potential MARCHF8 binding proteins. The top pathways involved in MARCHF8 interactions were analyzed by Ingenuity Pathway Analysis (F). MARCHF8 (G) and HLA-A/B/C (H) were pulled down from the cell lysate of SCC152 cells treated with MG132 using anti-MARCHF8 (G) and anti-HLA-A/B/C (H) antibodies, respectively, and analyzed by western blotting. All experiments were repeated at least three times.
Experimental models showed that removing or inhibiting MARCHF8 restored MHC-I expression and triggered a strong immune response against the tumours. This led to increased infiltration of immune cells, including CD8⁺ T cells, natural killer cells, and macrophages, within the tumour microenvironment. As a result, tumors that previously resisted treatment became susceptible to immune attack.
Single-cell RNA sequencing further revealed that loss of MARCHF8 reshapes immune interactions in the tumour microenvironment, enhancing cytotoxic immune activity. Importantly, combining MARCHF8 inhibition with existing immunotherapies transformed previously “cold” tumours into “hot” tumours that respond more effectively to treatment.
These findings suggest that targeting MARCHF8 could be a promising therapeutic strategy for improving immunotherapy responses in patients with HPV-positive head and neck cancers that are currently difficult to treat.
Journal article: Khalil, M.I, et al. 2026. The membrane-associated ubiquitin ligase MARCHF8 degrades MHC-I in HPV-positive head and neck cancer for immune evasion. PNAS.
Summary by Stefan Botha










