Chimeric antigen receptor (CAR) T cell therapy has shown limited benefit when combined with immune checkpoint inhibitors, and the reasons for this resistance have remained unclear. Recent research shows that CAR T cells struggle to maintain a population of stem-like T cells, which are important for sustained responses to therapies targeting the PD-1 immune checkpoint pathway (Figure 1).
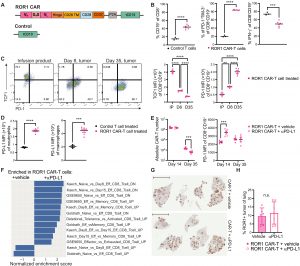
Figure 1: ROR1 CAR T cells fail to maintain a TCF1+ reservoir in lung tumors and are not enhanced by PD-L1 blockade. (A) Retroviral constructs used to engineer ROR1 CAR T cells and control T cells. Graphic created with BioRender (102). (B) Frequency (left), PD-1 and TIM-3 expression (middle), and IFN-γ production upon ex vivo restimulation with PMA and ionomycin (right) by infused CD8+CD19+ CAR T cells in tumors 8 days postinfusion into KPROR1 mice. n = 3 to 6 mice per group. (C) Flow plots (left) and quantification (right) of TCF1 and PD-1 expression by CD8+CD19+ CAR T cells over time in vivo. n = 5 mice per group with three technical replicates for IP. D8, day 8; D35, day 35. (D) PD-L1 expression on CD11b+Ly6G+ neutrophils and CD11c+F4/80+ macrophages in KPROR1 lung tumors 8 days postinfusion. n = 3 to 6 mice per group. (E) CD8+CD19+ CAR T cell numbers (left) and PD-1 expression (right) in KPROR1 tumors after treatment with vehicle or anti–PD-L1. n = 5 or 6 mice per group. (F) Normalized enrichment scores of gene sets significantly enriched (Padj < 0.05) in CD8 CAR T cells sorted from tumors of vehicle-treated or anti–PD-L1–treated KPROR1 mice 5 days postinfusion. n = 3 to 5 mice per group. (G) Representative immunohistochemistry (IHC) staining for ROR1 in KPROR1 mouse lungs 45 days post–CAR T cell infusion with or without anti–PD-L1 treatment. Scale bars, 5 mm. (H) Quantification of ROR1+ tumors from IHC images of KPROR1 lungs 45 days postinfusion. n = 6 or 7 mice per group. Data were analyzed by [(B), (D), (E), and (H)] unpaired Student’s two-way t test or (C) one-way ANOVA with Tukey’s posttest. Data are representative of two independent experiments. **P < 0.01, ***P < 0.001, and ****P < 0.0001. TM, transmembrane; tCD19, truncated murine CD19; MFI, median fluorescence intensity; ns, no significance.
In mouse models of lung cancer, CAR T cells rapidly lost the transcription factor TCF1, became functionally exhausted, and did not respond well to PD-L1 blockade alone. Increasing expression of the transcription factor c-Jun expanded stem-like CAR T cells within tumours, but PD-1 signalling still suppressed c-Jun at the post-transcriptional level, allowing exhaustion to persist.
However, combining c-Jun–engineered CAR T cells with PD-L1 blockade restored c-Jun expression, expanded CAR T cell populations, and led to near-complete tumour elimination. These findings suggest that modifying CAR T cells to preserve key transcriptional programs may improve their responsiveness to immune checkpoint therapies.
Journal article: Snyder, J.A., et al. 2026. Modulating AP-1 enables CAR T cells to establish an intratumoral stemlike reservoir and overcomes resistance to PD-1 blockade. Science Immunology.
Summary by Stefan Botha










