Never Too Early to Exercise: Long-Term Benefits of Early Life Exercise
22 April 2024
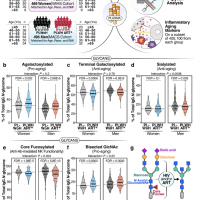
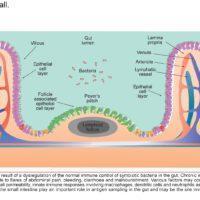
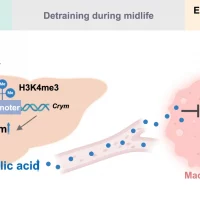
Researchers investigated the benefits of exercise on long-term health, particularly regarding whether the benefits of childhood exercise can have a lasting impact on adult health… Read More…